Note to the Reader This is a revised edition of a paper published in The Journal of Neuroscience in 1993. Several figures have been added. Text additions are in brackets [...].
Enlarging images. Thumbnail versions of all figures are embedded in the paper. Full-size images—usually under 300K—will download into a new window when you select the thumbnails. Drag the new window to the side of the text window. In some cases, high-resolution images—usually under 600K—that match the quality of the original micrographs can be downloaded by selecting the last line of the legend. These large images can be viewed with Adobe Photoshop, NIH Image, or equivalent.
The Journal of Neuroscience 13: 208–228 (1993)
Abstract Comparison of body and brain weights Evolutionary history of the domestic cat
Rapid Evolution of the Visual System: A Cellular Assay of the Retina and
Dorsal Lateral Geniculate Nucleus of the Spanish Wildcat and the Domestic
Cat
Robert W. Williams,1 Carmen Cavada,2 and Fernando
Reinoso-Suárez2
1Department of Anatomy and Neurobiology, College of Medicine, University
of Tennessee, Memphis, Tennessee 38163, USA and
2Departamento de Morfología, Facultad de Medicina, Universidad Autónoma de
Madrid, 28029 Madrid, Spain.
Contents
Introduction
Materials and Methods
Analysis of retinal structure
Analysis of ganglion cell numbers
Analysis of the LGN
Dwarfing
Domestication
Brain to body size scaling
ON- and OFF-center retinal ganglion cells
Development and brain evolution
Abstract
 The
large Spanish wildcat, Felis silvestris tartessia, has retained
features of the Pleistocene ancestor of the modern domestic cat, Felis
catus. To gauge the direction and magnitude of short-term evolutionary
change in this lineage, we have compared the retina, the optic nerve, and
the dorsal lateral geniculate nucleus (LGN) of Spanish wildcats and their
domestic relatives. Retinas of the two species have the same area.
However, densities of cone photoreceptors are higher in wildcat— over 100%
higher in the area centralis—whereas rod densities are as high, or higher,
in the domestic lineage. Densities of retinal ganglion cells are typically
20–100% higher across the wildcat retina, and the total ganglion cell
population is nearly 70% higher than in the domestic cat. These
differences are confined to the populations of beta and gamma retinal
ganglion cells. In contrast, the population of alpha cell is almost
precisely the same in both species. The wildcat LGN is much larger than
that of the domestic cat and contains about 50% more neurons. However,
cell size does not differ appreciably in either the retina or LGN of these
species. The differences in total numbers of ganglion cells and LGN
neurons correspond neatly to the overall decline in brain size in the
domestic lineage and to allometric predictions based on average species
differences in body size. We suggest that an increase in the severity of
naturally occurring cell death is the most plausible mechanism that can
account for the rapid evolutionary reduction in cell populations in this
feline lineage.
The
large Spanish wildcat, Felis silvestris tartessia, has retained
features of the Pleistocene ancestor of the modern domestic cat, Felis
catus. To gauge the direction and magnitude of short-term evolutionary
change in this lineage, we have compared the retina, the optic nerve, and
the dorsal lateral geniculate nucleus (LGN) of Spanish wildcats and their
domestic relatives. Retinas of the two species have the same area.
However, densities of cone photoreceptors are higher in wildcat— over 100%
higher in the area centralis—whereas rod densities are as high, or higher,
in the domestic lineage. Densities of retinal ganglion cells are typically
20–100% higher across the wildcat retina, and the total ganglion cell
population is nearly 70% higher than in the domestic cat. These
differences are confined to the populations of beta and gamma retinal
ganglion cells. In contrast, the population of alpha cell is almost
precisely the same in both species. The wildcat LGN is much larger than
that of the domestic cat and contains about 50% more neurons. However,
cell size does not differ appreciably in either the retina or LGN of these
species. The differences in total numbers of ganglion cells and LGN
neurons correspond neatly to the overall decline in brain size in the
domestic lineage and to allometric predictions based on average species
differences in body size. We suggest that an increase in the severity of
naturally occurring cell death is the most plausible mechanism that can
account for the rapid evolutionary reduction in cell populations in this
feline lineage.
[Key words: brain size, allometry, rods and cones, retinal ganglion cells, optic nerve, lateral geniculate nucleus, cell death]
Introduction
The tempo of mammalian brain evolution has been rapid. Twofold changes in
brain weight have occurred over periods of 1–5 million years in several
lineages, including that of humans (Edinger, 1948, 1966; Jerison, 1973,
1979; Radinsky, 1973, 1975, 1981; Eisenberg, 1981). Although this is an
interesting and important topic, it has been difficult to study the
process of brain evolution in any detail. Our approach has been to
identify a pair of closely related living species, one from a highly
conservative branch that has retained near identity with the ancestral
species, and the other from a derived branch that has undergone rapid
evolutionary change. The recent recognition that evolution and speciation
can occur in short bursts separated by long interludes of stasis provides
a sound theoretical basis for a search for such pairs (Schindewolf, 1950;
Eldredge and Gould, 1972; Stanley, 1979; Gould and Eldredge, 1986).
A candidate pair among mammals is the wildcat, Felis silvestris,
and the domestic cat, F. catus. Historical, biochemical, and
genetic data point to a derivation of the domestic cat from the North
African wildcat, F. silvestris lybica (Darwin, 1890; Clutton-Brock,
1981; Collier and O’Brien, 1985; Benveniste, 1985; Wayne et al., 1989).
This small North African wildcat descended from a larger ancestor whose
Pleistocene fossil record has been described carefully by Kurtén
(1965a,b). These Pleistocene wildcats share a constellation of
features—short premolar-canine diastema, large canine and carnassial
teeth, and a low crowned fourth lower premolar—with the particular
subspecies we have studied, F. silvestris tartessia (Kurtén,
1965a,b). In fact, the Spanish wildcat has more in common with wildcats
living toward the end of the Würm glaciation, 15,000–20,000 years BP than
with other extant F. silvestris subspecies (Table 1). For this
reason, Kurtén (1965b) concluded that the Spanish subspecies is an
isolated relict of an ancient Mediterranean wildcat population, that has
survived with minimal change for over 20,000 years in a warm Iberian
refuge.
We decided that a comparison of this particular wildcat subspecies
with the domestic cat could provide detailed insight into the range, rate,
and direction of evolutionary change in brain structure. For example, we
thought we would be able to determine whether the rapid decline in body
size in the cat lineage has been associated with a matched decline in
brain mass, and if so, whether this change has been brought about by a
reduction in neuron number or an increase in cell packing density. We
thought it would also be possible to determine whether numerical relations
among interconnected parts of the brain have been conserved in this
rapidly evolving mammalian lineage, and if so, then how precisely.
Finally, we thought it might be possible to compare ontogenetic
differences in these species and thereby expose developmental and cellular
mechanisms that ultimately underlie phyletic change.
Taking advantage of the wealth of data on the structure of the
primary visual system of domestic cats, we have begun with a quantitative
analysis of populations of cells in the retina and the dorsal lateral
geniculate nucleus of the Spanish wildcat. In this analysis we make two
explicit assumptions: first, that the adult wildcats we have had the very
rare opportunity to study are representative of the Spanish subspecies;
and second, that the Spanish wildcat is itself representative of, or at
least similar to, the ancestral population of Pleistocene wildcats from
which domestic cats ultimately trace their descent. The validity of these
key assumptions is supported by data summarized in the next section (Table
1). From this vantage point, we attempt to assess structural changes in
the primary visual system associated with approximately 25,000 years of
natural selection at the close of the Pleistocene, followed by 3,000–4,000
years of domestication.
Materials and Methods
Two adult wildcats (Felis silvestris tartessia Miller, 1907), a
male (S1) and a female (S2), were received from the Institute for the
Conservation of Nature (ICONA), Spain. Both animals had been rescued from
game traps in the Sierra Morena of south central Spain. The two animals
had been held for several months in a large outdoor pen. Breeding was
thought to have failed. Both animals were thin but in good health. They
both had the coat color and pattern typical of wildcats and many domestic
tabby cats (Haltenorth, 1953; Rodríguez de la Fuente, 1979). Each had four
prominent dark streaks running down the neck, a single broad dark band
running along the dorsal midline to the base of the tail, six to eight
less distinct dark arched bands running from back to abdomen, and three to
four dark rings around limbs and tails. Figure 26 in Haltenorth (1953)
shows a F. silvestris tartessia type specimen at the Zoologische
Staatssammlung (München, Germany) with a coat pattern almost
indistinguishable from that of our animals. The ages of the two wildcats
were unknown, but both appeared to be young adults. A single fetus was
found in the female after her perfusion.
Felis silvestris is not listed as an endangered or threatened
species under the U.S. Endangered Species Act or the Convention for
International Trade in Endangered Species of Wild Fauna and Flora (CITES,
1991). However, all felidae except the domestic cat, Felis catus,
are listed in Appendix 2 of CITES and are subject to import and export
regulations. The wildcat is protected by law in Spain. We complied with
all conditions of the ICONA permit and all relevant CITES regulations.
Table 1. Size of teeth used to assess status of wildcats (click to
magnify)
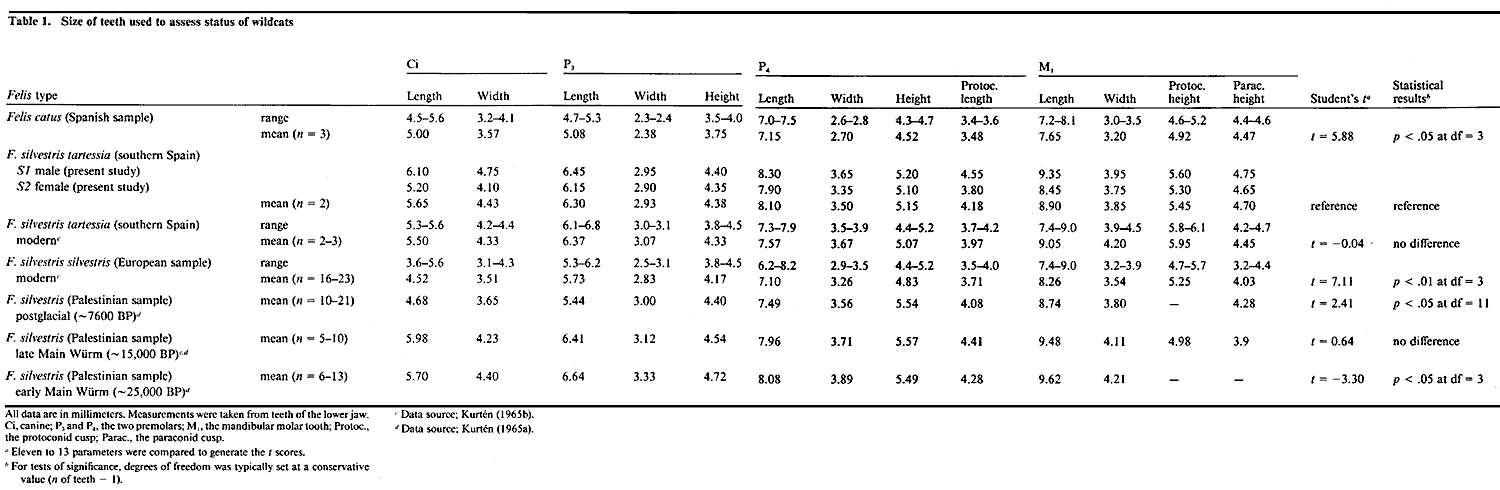
Confirmation of the wildcat subspecies and the morphological similarity between Spanish wildcats and Pleistocene wildcats. The size and morphology of the teeth and jaws are among the most reliable criteria by which to identify species and subspecies of vertebrate (Kurtén 1954; Carroll, 1988). Without resorting to statistical analysis, the dental measurements taken from our specimens conform closely to those of three F. silvestris tartessia specimens previously studied by Kurtén (1965a). What is even more remarkable and significant is that our wildcats also do not differ appreciably from a sample of Pleistocene wildcats dated approximately 15,000 years before present (late Main Würm). A relatively objective index of the degree of similarity or dissimilarity is provided by the t value calculated for all 13 parameters (values to the right of Table 1). This more rigorous comparison demonstrates that our Spanish wildcats are remarkably similar to Late Main Würm wildcats (t = 0.64), but differ greatly from modern European wildcats (t = 7.11), from domestic cats, including a huge 9 kg domestic tabby cat (t = 4.76), and from a sizable sample of Neolithic (7,500 BP) wildcats (t = 2.41). The overall body size of our wildcats also overlaps measurements on F. silvestris tartessia provided by Haltenorth (1953, his Tables 2 and 4). In sum, the animals we have studied have been correctly classified as F. silvestris tartessia. They are certainly not feral domestic cats, wild hybrids, or a different subspecies of Felis silvestris. The statistical comparisons of data in these tables also document the striking similarity between the Spanish wildcats we studied and wildcats at the close of the Pleistocene, approximately 15,000 years BP. For example, a comparison of the 13 parameters listed in Table 1A gives a Student’s t value of merely 0.64—an insignificant difference. In contrast, the Spanish wildcat differs significantly from other modern European wildcats (t = 7.11). data also provide a strong case that Felis silvestris tartessia represents an isolated relict of a late Pleistocene wildcat population.
Sources of domestic cat material. Domestic cat tissue was obtained from our colonies in Madrid and the University of Tennessee and from Drs. L. M. Chalupa, A. Elberger, and D. Frost. An extremely large domestic cat was provided to us by Dr. A. Tuberville to explore the upper limits in the size of the brain and dentary that might be expected among domestic cats. This huge young domestic cat weighed 9 kg, was 61 cm from crown to rump, and had femurs that were 16.5 cm long. Femurs of European wildcats are typically less than 13 cm long (Rohrs, 1955).
HRP injection procedure. Injections of horseradish peroxidase (HRP) were made into the left dorsal lateral geniculate nucleus (LGN), left superior colliculus, and left pretectal region of the male wildcat (S1). Similar unilateral (n = 1) and bilateral (n = 3) series of injections were made into normal domestic cats. Some of the domestic cat retinas have been described and illustrated in previous studies (Chalupa et al., 1984; Kirby and Chalupa, 1986). Animals initially received a single intramuscular dose of ketamine (0.4 cc) and atropine (0.2 cc) and were subsequently anesthetized with an intravenous injection of sodium pentobarbital (30 mg/kg). They were placed in a stereotaxic device and craniotomies were made over the approximate locations of the left superior colliculus, pretectum, and dorsal lateral geniculate nucleus. The boundaries of retinorecipient nuclei were mapped by recording multicellular visually evoked activity with a tungsten microelectrode. A series of 150–200 nl injections of 25% HRP diluted in pure dimethylsulfoxide were made along each penetration within the LGN (25 penetrations), pretectum (6 penetrations), and superior colliculus (10 penetrations) as described in Chalupa et al. (1984). The total volume of HRP delivered in a single penetration ranged from 200 to 600 nl.
Perfusion and dissection of the animals. All animals received an
overdose of pentobarbital (200 mg) and were subsequently perfused through
the heart with phosphate buffered saline (0.1 M, pH 7.3), followed by a
mixture of 1.25% paraformaldehyde and 2.5% glutaraldehyde in phosphate
buffer. The cranial vault was removed in one large intact piece, and the
brain—including olfactory bulb, pituitary, and the entire medulla (Fig.
1)—the eyes, and segments of most cranial nerves were removed. Retinas
were taken from the eyes and flattened by making a set of radial cuts. The
areas of flatmounted retinas were measured before dehydration. Crania and
dentaries were cleaned in a 25% solution of bleach. Pelts of both wildcats
were returned to ICONA.
Following the transcardial perfusion of wildcat S2, an adult female
that we had palpated on two previous occasions to be certain that she was
not pregnant, we explored the abdominal cavity and found a single fetus.
The fetus was immediately removed and fixed briefly by transcardial
perfusion followed by prolonged immersion in fixative. Preservation of
optic nerves proved to be surprisingly good, and given the value of this
rare fetal wildcat material, we undertook a detailed analysis of both
nerves using procedures identical to those described in a previous study
on the optic nerves of domestic cat embryos (Williams et al.,
1986).
Processing of retinas. A modification of the Hanker-Yates procedure (Perry and Linden, 1982; Chalupa et al., 1984) was used to demonstrate the presence of HRP within retinal ganglion cells in wildcat S1 and normal domestic cat retinas. The retinas of the female wildcat S2 were stained with toluidine blue using the method described by Wong and Hughes (1987a). Additional domestic cat retinas were mounted between coverslips in glycerin and used for the analysis of the photoreceptor mosaic using methods summarized in Williams (1991).
Brain histology. Brains were hemisected in the sagittal plane. The left hemispheres were cut frozen at 50 µm in the coronal plane. These frozen sections were collected and distributed in several series. The first and third series were stained with cresyl violet; the second series was in some cases reacted for HRP histochemistry (S1, D1)using diaminobenzidine as the chromogen. Right hemispheres were embedded in celloidin and cut in either the coronal or the horizontal (S2) plane at 50 µm. For comparison, frozen and celloidin coronal sections from 11 domestic cats cut frozen (n = 7) or embedded in celloidin (n = 4) in the coronal plane were were examined.
Photoreceptor analysis. The density of rods and cones was measured along the horizontal meridian in the left retina of S1 and two domestic cats. A x63/1.25 N.A. semiapochromatic objective with a very long working distance (500 µm) was used to focus on the photoreceptor mosaic from either the vitreal or scleral side of the retina using differential interference contrast (DIC) optics, a video overlay system, and semiautomatic image analysis software written for use with an Apple Macintosh computer (Fig. 2). A 50-watt high-pressure mercury light source was used with or without narrow-band interference filters to obtain optimal contrast and resolution. Analog and digital video enhancement were used to improve the contrast of the photoreceptor mosaic as viewed on an RGB monitor. Rods and cones were counted in fields of 144 µm2 and 1000 µm2, respectively, using a video overlay setup (Wikler et al., 1990; Williams, 1991). Locations of sites in the retina were measured with reference to the center of the area centralis using linear encoders attached to the stage (Heidenhain Inc., LS 403 encoders, 0.2 µm resolution, accuracy ± 10 µm over 50 mm).
Retinal ganglion cell analysis. The analysis of the distribution
and types of retinal ganglion cells was also carried out using DIC optics.
The depth of ganglion cell dendrites was measured with reference to the
inner border of the inner nuclear layer or the equator of ganglion cell
bodies using a linear encoder (Heidenhain M25, 0.1 µm resolution, accuracy
0.3 µm over 25 mm). The tip of this encoder rested on the stage.
High-magnification plots of ganglion cells (e.g., Fig. 7) were made by
overlaying the video image of the retina onto a graphics program with
variable magnification as shown in Figure 11F. The coordinates of ganglion
cells on these plots are accurate to within 10 µm. Low-power plots were
made using a drawing attachment and a x4 planapochromatic objective.
The classification of retinal ganglion cell types (on-center versus
off-center; alpha, beta, and gamma cell types) was carried out using a
x100/1.25 N.A. planachromatic objective and DIC optics. On-center and
off-center cells have dendritic arbors in the inner and outer parts of the
inner plexiform layer, respectively (Peichl and Wässle, 1981; Wässle et
al., 1981a,b). With one or two exceptions, all alpha and beta cells in
regions chosen for analysis could be confidently classified. For every
cell that we categorized as on-center or off-center, we first measured the
distance (depth) from the equator of the ganglion cell to the position of
secondary and tertiary dendrites in the inner plexiform layer. The
accuracy of these measurements was about 0.5 µm. Gamma cells could
generally not be classified due to their poor dendritic labeling.
Similarly, cells within 2 mm of the area centralis could not be identified
reliably due to poor dendritic filling. In classifying cells in the
wildcat we have relied heavily on criteria developed in the domestic cat
(Boycott and Wässle, 1974).
For the high-resolution sampling of ganglion cell densities in the
area centralis and along the horizontal meridian, the distance between
adjacent sample sites varied from 35 µm in the area centralis to 200 µm in
the far periphery. Sites were studied with a x100/1.25 N.A. planachromatic
objective. Each site had an area of 1200 µm2.
Sampling and counting procedures were unbiased (Brændgaard and Gundersen,
1986), and no adjustments were made to avoid blood vessels or axon
fascicles or small tears in the wholemount. For the purpose of counting,
the nucleolus was used as the criterion structure. Data were collected
separately for labeled and unlabeled cells in the ganglion cell layer.
Glial cells, characterized by their small cell bodies and minuscule
nucleoli (Wong and Hughes, 1987b), were excluded from analysis. To
determine peak densities of ganglion cells in the area centralis,
video-enhanced DIC optics were used to analyze sets of highly magnified
fields (450 µm2 at x6500). The highest average
of six adjoining fields was used to calculate the peak density of ganglion
cells.
Numbers of axons in the optic nerve. Cross-sections of the
mid-orbital part of the optic nerve were prepared for analysis as
described in previous work (Williams and Chalupa, 1983; Williams et al.,
1986). Counts of axons were obtained in a systematically distributed
sample of fields (typically 50 to 100), and our estimate of average axon
packing density was multiplied by the cross-sectional area of the nerve to
obtain an estimate of the total axon population. A change in sampling
protocol was required because of the large area of the wildcat optic
nerves. The ultrathin sections of the wildcats were too large to fit onto
single grids. Consequently, several grids had to be photographed to sample
the entire cross-section of the nerve. Electron micrographs covered fields
of 467 µm2. The sample area was determined with
the aid of a mesh calibration grid photographed at the end of the sampling
session. Axons in cats are unbranched and therefore the number of optic
axons provides a good estimate of the retinal ganglion cell population (Chalupa
et al., 1984; Lia et al., 1986).
In the case of the fetal wildcat, a total of 20 to 25
systematically distributed fields, each with an area of 93 µm2,
were photographed and counted in each optic nerve. Numbers of axons,
growth cones, and necrotic fibers were counted in each field, using the
same criteria developed in Williams et al. (1986,
1991).
LGN cell analysis. The total population of neurons in the
LGN was estimated in both frozen and celloidin embedded material by means
of direct three-dimensional counting using oil immersion objectives and
DIC optics (Williams and Rakic,
1988b). The
main advantage of three-dimensional counting is that no correction
factors—particularly, the often inaccurate Abercrombie correction—are
required to estimate local cell density. Furthermore, this method is
insensitive to variation in processing methods and section thickness.
Neurons in the LGN were distinguished from glial cells and endothelial
cells on the basis of size and nuclear staining as described in detail in
Williams and Rakic (1988a).
Neuronal nuclei, both of principal neurons and interneurons, were counted
in translaminar probes extending through layers A, A1, C, C1, C2, and C3
(and the medial interlaminar nucleus in the most medial probes). Between 9
and 11 probes were made through each nucleus in regions representing
upper, lower, central, and peripheral visual fields. Each probe was made
up of a series of adjacent counting boxes. The depth of the counting
box—the z-axis of the section—was optimized for the thickness of the
individual section. The tops and bottoms of counting boxes were defined
using the linear optical encoder. The average density of neurons in the
counting boxes was calculated and multiplied by the volume of the nucleus
to obtain the total LGN neuron population.
The volume of the LGN was determined by direct integration. To do
this the areas of 8 or more sections through each nucleus were measured
and plotted against the rostral-caudal (or, in some cases, dorsal-ventral)
position of the section. A curve was fit by eye through the points, and
the area under this curve—an accurate estimate of total LGN volume—was
determined. Although this method is not as simple as several alternatives
described in Rosen and Harry (1990), it provides a useful graphic
assessment of the shape of the nucleus, and it allowed us to detect and
correct errors made during drawing and measuring the LGN cross-sections.
The size distribution of neurons in the LGNs of wildcats and
domestic cats was studied in celloidin-embedded tissue. The
cross-sectional areas of cell bodies were measured directly onscreen at
x2000 using a video-enhanced DIC overlay system (Williams and Rakic,
1988a) through layers A, A1, and the C layers. A 100 µm graticle divided
in 2-µm increments was used to calibrate all images.
Results
We have studied two adult wildcats and one fetal wildcat. These animals were extremely difficult to obtain and there was no immediate prospect for increasing numbers of cases. Therefore, our approach has been to analyze the material in detail, as if we had uncovered a small number of rare and important fossils. In some cases, our analysis of the wildcats has extended beyond data initially available for the domestic cat. This is true for the analyses of photoreceptor distribution and dendritic differences among on-center and off-center beta cells. Despite the small sample size, the central result of this study—the marked differences in populations of retinal ganglion cells and of geniculate neurons—do achieve statistical significance. With respect to other facets of this study, particularly those based on single animals, we have tried to be circumspect in our conclusions and to point out interpretative caveats. In several instances we provide more complete documentation for the wildcats than for domestic cats. References to specific figures for domestic cats are made both in the text and in figure captions.
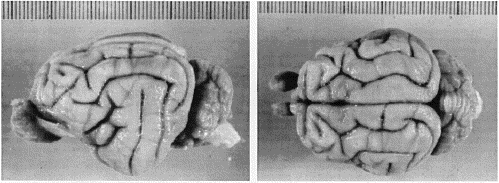
Figure 1. Dorsal and left lateral views of the brain of
Felis silvestris tartessia, the Spanish wildcat (female S2). Sulcal
patterns in this 31-gm brain conform to the type III pattern seen in about
18% of domestic cats (fig. 7 in Otsuka and Hassler, 1962).
Comparison of Body Weight and Brain Size of the Two Species
The average body weight of F. silvestris tartessia males is about 6.5 kg, and of females about 4 kg (Rodríguez de la Fuente, 1979). Our two wildcats were lean and weighed less than average: the male (S1) weighed 3.7 kg and the female (S2) weighed 3.3 kg. Despite these modest body weights, the brains of the male and female wildcats weighed 37 gm and 31 gm, respectively. These values are far above the domestic cat average: 27.6 gm with a standard deviation of ± 1.5 gm for male domestic cats, and 26.5 ±1.35 gm for females (Latimer, 1938). Even the brain weight of the small female wildcat reproduced in Figure 1 is 3.3 standard deviations above that of female domestic cats. Our huge 9-kg male domestic cat had a brain weight of only 28.25 gm. This is 9 gm less than the brain weight of the less massive but equally tall male wildcat. In absolute terms, the difference in brain weight between the two adult wildcats and domestic cats is in the neighborhood of 20 to 30%. This difference conforms closely to that which we anticipated based on the allometric relationship between brain and body weight derived from a wide range of felids (Davis, 1962; Radinsky, 1975; Pagel and Harvey, 1989):
brain mass = 0.23 (average body mass)0.61
The pattern of gyri and sulci in wildcats conforms to the typical felid pattern (Fig. 1; cf. Radinsky, 1975). The lateral, posterolateral, and suprasylvian gyri are arranged in a common pattern that Otsuka and Hassler (1962) refer to as type III. The lateral and suprasylvian gyri are wider in the wildcats than is typical in domestic cats. For example, the lateral gyrus was between 7.0 and 8.5 mm wide in the wildcats compared to typical values between 5.0 and 6.5 mm in domestic cats (in vivo values, Reinoso-Suárez, 1961).Variation in the sulci in wildcats and domestic cats, particularly in the occipital region, has been reported previously by Röhrs (1955). However, given the extensive variation in sulcal patterns in domestic cats (Otsuka and Hassler, 1962; Kawamura, 1971), and even between right and left sides of one of our wildcats (Fig. 1), such differences are not reliable criteria for distinguishing between species. Although the breadth of the cranium is greater in wildcat, the stereotaxic coordinates of bregma and inion do not differ appreciably between species. The thickness of bones of the cranial vault is much reduced in wildcats (for example, the thickness of the temporal bone is merely 1.25 mm in wildcat versus 2.6 mm in domestic cats). In this case, the thick skulls of domestic cats do house smaller brains.
Analysis of Retinal Structure
Constancy of retinal dimensions. There is complete overlap in the area of wildcat and domestic cat retinas. The range among the four wildcat retinas is from 528 to 570 mm2, whereas the range among domestic cats is from 460 to 640 mm2 (our data; Hughes, 1975; Chalupa et al., 1984). Our huge domestic cat had retinal areas of 570 and 600 mm2. Linear retinal measurements are also equivalent: the distance from the center of the area centralis to the center of the optic disk in the wildcat retinas is between 3.37 mm (S2 right) and 3.55 (S1 right), precisely in the same range as that reported in Nikara et al. (1968), Wässle et al. (1975), and Hughes (1975). It is also likely that posterior nodal distances and retinal magnification (about 213 µm per degree near the area centralis) in the two species are the same.
Differences in rod and cone distributions . A quantitative analysis of the photoreceptor mosaic was possible in both retinas from the male wildcat S1. Data from this animal demonstrate that densities of cone photoreceptors can reach higher densities in wildcats than has ever been reported in domestic cats. The difference appears to be most marked in the central retina, particularly at the center of the area centralis where cone densities of up to 100,000/mm2 were encountered in this wildcat (Fig. 2). In comparison, the highest cone density we have yet encountered in domestic cats is between 35,000/mm2 and 40,000/mm2, a range that is itself about 25% higher than peak cone densities reported in previous studies of this species (Steinberg et al., 1973, their figs. 1, 2, and 6; Wässle and Riemann, 1978, their fig. 2). Cone densities are also high in the periphery of this wildcat’s retinas (Figs. 3, 4). However, the difference is not as great as in the area centralis. For example, along most of the horizontal meridian, cone densities are only 20–30% higher than in the domestic cat at comparable eccentricities. Although not studied in the same detail, there also appears to be a comparative surplus of cones in dorsal and ventral retina of the wildcat in comparison to the domestic cat. To obtain a rough estimate of the difference in the total cone population, we integrated the area under the curves in Figs. 3 and 4. The ratio of these areas is 1:1.3 (domestic:wildcat). Therefore, along this axis, domestic cats typically have 25% fewer cones than does wildcat S1.

Figure 2. The photoreceptor mosaic and ganglion cell layers of the
male wildcat, S1. All images cover an area 78 by 100 µm. A and B
are pairs of differential interference contrast images of precisely the
same area of a retinal wholemount, 0.5 mm above the area centralis close
to the decussation line in temporal hemiretina (ipsilateral to the HRP
injections). The two micrographs were taken at focal planes separated
vertically by 74 µm. A is at the level of the photoreceptor inner
segments. Large cone inner segments stand out clearly among the small rod
inner segments. A single cone is marked by a small arrowhead. This image
can be compared to those reproduced in figure 2 of Steinberg et al.,
(1973) and figure 2 of Wässle and Riemann (1978). B is a micrograph
taken at the level of the ganglion cells. Most of these cells are labeled
with the dark HRP reaction product, but at least one unlabeled cell with a
large nucleolus is also a ganglion cell (arrowhead), presumably one that
has a crossed projection. C and D are comparable
through-focus pairs of micrographs taken in the area centralis. Note that
the cones in area centralis are somewhat smaller than in A and are
much more densely packed (densities range from 50,000 to 100,000). Smaller
rods are also scattered throughout the area centralis. We have not been
able to find regions of such high cone density in domestic cats, nor have
other investigators (see Steinberg et al., 1973, their figure 2). In D,
only a single labeled ganglion cell is present in this field just nasal to
the decussation line in the retina ipsilateral to the central HRP
injections. Only 49 other ganglion cell bodies are visible in this focal
plane. However, ganglion cells are stacked 2 or 3 cells deep here and this
small field actually contains 112 ganglion cells. This is equivalent to a
density of 14,400 cells/mm2.
Conversely, the rod densities were much lower in the area centralis of wildcat S1 than has ever been measured in domestic cats (our data and Steinberg et al., 1973). Rod densities drop to well under 50,000/mm2 in the center of the area centralis of S1 (Fig. 2), but in domestic cats we have been unable to locate any fields in the area centralis of any animal in which rod densities fall to less than 200,000/mm2. Along most of the horizontal streak of the wildcat, rod densities average between 300,000 and 400,000/mm2. This is on the low side of the range we have observed in the horizontal streak of domestic cats. In both species, rod densities range up to 550,000 in the dorsal periphery. To estimate the difference in the rod population we again integrated the area under the curves (Figs. 3 and 4). The ratio is 1.2:1 (domestic:wildcat).
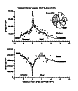
Figure 3. Gradients in wildcat cone and rod densities through the
area centralis and along the horizontal axis (male wildcat S1). The
vertical axes in this figure and figure 4 (domestic cat) are identical
except that the y axis is interrupted above 30,000 mm2.
The gray bars at the far right and left of these plots represent
eccentricities at which we could not make accurate measurements because of
excessive retinal pigment. A sketch of the ipsilateral HRP-labeled retina
in the upper right shows the horizontal line along which data were
collected.
Figure 4. Gradients in domestic cat cone and rod densities through the area centralis and along the horizontal axis (domestic cat D8). Faint gray curves in both A and B are taken from data in Steinberg et al. (1973).
Marked Differences in Ganglion Cell Numbers and Distribution
Total ganglion cell densities are substantially higher in all retinas of both wildcats than in domestic cats, both in the area centralis and across the retinal periphery (Fig. 5A,B). For example, in the area centralis, ganglion cell densities are 50% to nearly 100% higher in the wildcats than in domestic cats. In particular, the peak density of HRP-labeled ganglion cells in the right retina of S1 (contralateral to the injected hemisphere) is 15,100 cells/mm2 at a location 40 µm nasal to our estimate of the position of the center of the decussation line (Fig. 6A,B). At the decussation line itself, the density of labeled and unlabeled cells in S1 is 10,100 and 5,900 cells/mm2, respectively, giving a peak density of about 16,000/mm2. In wildcat S2, a case in which both retinas were stained with toluidine blue, the peak density of cells with large nucleoli in the area centralis is 18,400 in the left retina (Fig. 6D) and 20,900 (a peak of 26 cells in an area of 1250 µm2) in the right retina. In comparison, the highest packing density of HRP-labeled retinal ganglion cells we have found in the center of the area centralis of a domestic cat is 10,500/mm2 (Fig. 5B). This value matches peak ganglion cell densities reported in numerous previous studies of domestic cat retina (Hughes, 1975; Stone, 1978; Stone et al., 1982; Chalupa et al., 1984; Wong and Hughes, 1987a). It is probable that the reduction in cone and ganglion cells densities in the area centralis of the domestic lineage have been matched by a reduction in sampling resolution of both cell arrays, as well as in photopic acuity. Ganglion cell densities in the periphery are also greater in both wildcats than in domestic cats. However, the quantitative difference is less marked, averaging of 15-25% (Fig. 5). For example, in the field of cells depicted in Fig. 7, the density of HRP-labeled ganglion cells is 300/mm2 at a mean eccentricity of 9.4 mm. At this eccentricity in the domestic cat, ganglion cell densities are typically approximately 200/mm2 (Fig. 5B, 8, and see Stone, 1978; Chalupa et al., 1984).
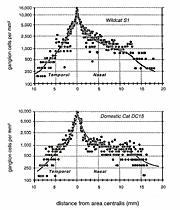
Figure 5. Central-to-peripheral gradient of ganglion cell density
along the horizontal axis in wildcat S1 (A) and domestic cat, DC15
(B). Qualitatively, the gradients are the same in both species, but
densities are higher, most prominently, around the area centralis of the
wildcats. To obtain the plot in A, numbers of HRP-labeled retinal
ganglion cells were counted and summed in both retinas ipsilateral and
contralateral to the series of unilateral HRP injections (see Fig. 6).
Data were sampled in a completely unbiased manner and include fields
traversed by large blood vessels. This accounts for some of the
variability among data points.
Decussation pattern does not differ. The pattern of decussation of ganglion cell axons was examined in wildcat S1 by retrograde labeling from one hemisphere (Fig. 6A, B). The characteristics of the decussation pattern and the relative sizes of the temporal and nasal retinal components appear indistinguishable at a qualitative level from those noted in domestic cats by previous investigators (Cooper and Pettigrew, 1979; Jacobs et al., 1984, their figure 1).
The horizontal streak. We were struck by the prominence of the
horizontal streak in the wildcat retinas. However, our quantitative
analysis revealed that the increment in cell density in the wildcat streak
is of the same magnitude as that seen elsewhere in peripheral retina (Fig.
5). For example, the density of labeled retinal ganglion cells in the
mid-nasal periphery of the visual streak (6–12 mm eccentricity) averages
1,000 cells/mm2 in wildcat S1 and about 600–700
cells/mm2 in domestic cats (Rowe and Stone,
1976; Chalupa et al., 1984). Studies by Peichl and colleagues (L. Peichl,
personal communication) have shown a marked difference in the development
of the horizontal steak in wild and domestic dogs.
Given the lower densities of ganglion cells in the domestic cat,
most prominently around area centralis, one might expect a compensatory
increase in the spread of dendritic fields in order to conserve field
overlap (Wässle et al., 1981a). To answer this question unequivocally
would require more complete dendritic filling than we have been able to
achieve in either beta or gamma cell classes. However, the marked species
differences in ganglion cell density at the area centralis and the sharp
peak in cone density in wildcat S1, suggest that visual acuity is higher
in wildcat than domestic cat.

Figure 6. The area centralis of the wildcat retina and the
precision of the line of decussation. A. the central region of the
retina contralateral to hemispheric HRP injections in the male wildcat S1.
B. The ipsilateral left retina from the same animal. The regions
shown in A and B are 1.08 mm by 1.43 mm. For alignment
purposes, the left retina in B has been oriented so that dorsal is
down. In the wildcat, as in the domestic cat, there are numerous cells in
temporal hemiretina that have crossed projections (left side of
A), but there are very few cells in the nasal hemiretina that have
uncrossed projections (right side of B). Comparable figures
of the line of decussation in domestic cat are provided in Jacobs et al.
(1984, their figure 1). C. Higher magnification micrograph of the
center of the area centralis. Note the relatively abrupt change in
patterns of decussation as revealed by the degree of overlap of labeled
and unlabeled retinal ganglion cells. Arrowheads in A and
C point out a common landmarkùa large alpha cell. Magnification in
C is x342. D. The entire area centralis in a Nissl-stained
preparation from wildcat S2. The large darkly stained cells are alpha cell
bodies. Alpha cells in the center of the area centralis do not stain as
darkly, in part because the ganglion cell layer is comparatively thick.
The region of the wildcat area centralis in which ganglion cell densities
are above 10,000/mm2 is small, corresponding
approximately to a circle with a radius of 150 µm (see Fig. 9). In this
zone, the ganglion cell layer is typically two cells thick. Magnification
in D is x72 and the field is 1.36 mm by 1.25 mm in horizontal and
vertical dimensions.
Conservation of alpha cell number and distribution. While the total ganglion cell density is higher in wildcats than has ever been reported in the domestic cat, this is not true of one particular ganglion cell class: densities of alpha cells are almost precisely the same in both species (Fig. 9). For example, alpha cell density in the area centralis is between 120 and 150 cells/mm2 in both wildcats, whereas in domestic cats, alpha cell density is between 130 and 200 cells/mm2 (our data from three domestic cats; cf. Wässle et al., 1975; Stone, 1978; Kirby and Chalupa, 1986). We counted 410, 425, and 428 HRP-labeled alpha cells in a 3-mm-high by 2-mm-wide region located just nasal to the area centralis in three domestic cats. In comparison, we counted 411 HRP-labeled alpha cells in the same area in wildcat S1. Similarly, in the HRP labeled retina of wildcat S1, in which alpha cells can be identified unambiguously even in the center of the area centralis, there are 53 alpha cells in a nasal hemicircle with a radius of 500 µm centered on the area centralis (52 labeled and 1 unlabeled). This is the precisely the same number of alpha cells as identified in this region by Wässle et al. (1975, their fig. 8A). Similarly, we identified a total of 1,412 Nissl-stained alpha cells in the 36-mm2 region centered on the area centralis in wildcat S2 (Fig. 9), while Wässle et al. (1975, their figures 6A and 8A) identified 1,484 alpha cells in an equal area in a domestic cat—a difference of merely 5% (data derived from their figs. 6A and 8A). Densities of alpha cells are also remarkably close in the mid and far periphery of both species. For instance, the density of HRP-labeled alpha cells 9-10 mm above the area centralis in dorsal retina just nasal to the decussation line is 14/mm2 in wildcat S1, and 11/mm2 in wildcat S2. In domestic cats, values in this region range between 10 and 13/mm2 (Wässle et al., 1975, their fig. 6A; Stone, 1978, his fig. 5). Thus, it seems highly probable that the total alpha cell population in wildcats is in the same range as that for the domestic cat—5,000 to 7,000 (Wong and Hughes, 1987b).
Figure 7 (large). Retinal ganglion cell mosaic in the wildcat S1. This field of cells covers an area of about 1 mm2 in the dorsal part of the retina. Coordinates are indicated on the axes. Large off-center alpha cells are represented by black irregular shapes. On-center alpha cells are similar, but unfilled. On- and off-center beta cells are represented by the large circlesùblack for off-center, unfilled for on-center, and gray for two beta cells of unknown subtype. The small circles represent the heterogeneous gamma cell class. Only a few of these cells could be categorized as on-center or off-center. Four unlabeled small neurons were identified in this field. They may have been unlabeled gamma cells or displaced amacrine cells.
No decline in alpha cell density in the wildcat’s area centralis. In wildcat S1 the pattern of HRP-labeled cells allowed us to define the location of the center of the area centralis with a precision of ± 25 µm, independent of the alpha cell distribution itself (Fig. 6A, B). This made it possible to assess whether or not there is a local decline in alpha cell density in the wildcat area centralis—an unresolved issue that has arisen in the domestic cat retina (Wässle et al., 1975; Stone, 1978; Mountcastle 1980, p. 539). In the wildcat, the density of alpha cells within a radius of 100 µm of the center is 127/mm2 and in two slightly more peripheral annuli, densities are 121 and 132 alpha cells/mm2. Nor was a central decline in alpha cell density noted in S2 (Fig. 9). Similar analyses of HRP-labeled retinas from three domestic cats, also did not demonstrate any appreciable decline in alpha cell density at the precise center of the area centralis.

Figure 8
(large). Retinal ganglion cell mosaic in the domestic cat at a
comparable location to that shown in fig. 7 for the wildcat. All
conventions as in figure 7. The dendritic arbors of alpha cells have been
drawn somewhat more completely in this plot than in that shown for the
wildcat.
Relative abundance of alpha, beta and gamma ganglion cells are
similar. The numbers of the alpha, beta, and gamma cell types were
studied in the dorsal periphery of wildcat S1. Here the proportions of HRP-labeled
ganglion cell types are similar to those in domestic cats (Figs. 7, 8).
Beta cells make up 50.2% of the local ganglion cell population (155 of
309), gamma cells make 45.3%, and alpha cells make up the remainder (14 of
309 cells in an area of 1.08 mm2). In the
domestic cat, beta cells make up 48.9%, gamma cells make up 45.3%, and
alpha cells make up the remainder (13 of 225 cells in an area of 1.08 mm2).
Thus, ratios of cell types in a region of retina in which the assignment
of cell types is unambiguous are similar in the two species. However,
given the finding that the alpha cell population is the same in both
species, but that the total population of ganglion cells is higher in
wildcats, it is clear that the overall proportion of alpha cells must be
slightly lower in wildcats than is typical in domestic cats (cf. Stone,
1978; Wong and Hughes, 1987b). Furthermore, at the area centralis of the
wildcat, a region in which the total ganglion cell density is particularly
high, alpha cells make up a smaller percentage (<1%) of the cell
population than in domestic cats (2–3%).
As has also been noted in the domestic cat (Wässle et al., 1981a,
and see our Fig. 8), there are slightly more off-center beta cells than
on-center beta cells (26.9% off-center, 22.7% on-center, 0.6% unknown) in
the wildcat dorsal periphery (Fig. 7). Corresponding percentages for the
domestic cat are 26.2% off-center and 22.7% on-center cells (Fig. 8).

Figure 9. Alpha cell distribution in and around the area centralis
of wildcat S2. The center of the area centralis, a region is which total
ganglion cell densities in wildcat are above 10,000/mm2
and in which cone densities are elevated, is encircled. The region
reproduced in Fig. 6D is outlined by the solid rectangle. Comparable data
for alpha cell density in central retina of the domestic cat are provided
in figure 8A of Wässle et al., (1975).
Ganglion cell size is the same. The higher average cell density in the wildcat retina could be associated with a reduction in mean cell body size. Such changes have been found following experimental manipulations in domestic cats (Kirby and Chalupa, 1986). We analyzed the sizes of all the cells in Figures 7 and 8. In wildcat S1, the average cross-sectional area of 309 ganglion cells at a location 9 mm above the area centralis is 357 µm2 (equivalent to an average diameter of 21.3 µm). In the domestic cat, the average size of the 230 ganglion cells in a comparable region, plotted in Fig. 8, is almost precisely the same—359 µm2. This equality in ganglion cell size is remarkable given the sizable difference in the local density of ganglion cells—286 cells/mm2 in the wildcat and 208/mm2 in the domestic cat.
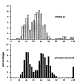
Figure 10. Ganglion cell size in a wildcat and two domestic cats at
sites 9 mm above the area centralis in the nasal hemiretina. HRP labeled
cells were measured. Note the substantial variation in the two lower
histograms taken from two domestic cats.
More subtle differences in cell size between species are, in general, not masked by averaging across cell classes (Fig. 10). For example, the average beta cell diameter in S1 and the domestic cat DC13 are both 23 µm. Similarly, the average diameters of gamma cells in S1 and DC12 are both about 15 µm. However, cell size is quite variable among domestic cats, even at equivalent retinal coordinates. For instance, in domestic cat DC12 the mean size of HRP-labeled retinal ganglion cells 9-10 mm above the area centralis was 472 µm2 versus 359 µm2 in another domestic cat, DC13. It is also the case that HRP-labeled alpha cells in the wildcat S1 are smaller than in the domestic cats (Figs. 7, 8, 10). Given the small sample size, it seems prudent simply to conclude that data on cell size in wildcat S1 falls within the range seen in domestic cats.
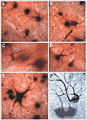
Figure 11
(large). On-center and off-center ganglion cells in the wildcat.
A. Three off-center beta cells (arrowheads) have dendrites located
in the inner plexiform layer in a focal plane 12 µm away from the location
of their cell body centers. B. Typical thin dendrites of two
adjacent on-center beta cells located 5 µm distant from the center of
their cell bodies. C. A single field photographed at two focal
planes in the inner plexiform layer. The upper half of the field is filled
by the large dendrites of an off-center beta cell at a depth of 15 µm. The
lower half shows part of the arbor of an on-center beta cell at a depth of
8 µm. This field is located 2.5 mm dorsal to area centralis. D.
Typical asymmetrical orientation of off-beta cell dendrites. Note the
large caliber of the dendritic trunk. E. Large on-center alpha
cell, 6 beta cells and 3 gamma cells. The identity of each cell in this
field is depicted schematically in fig. 7 at a location 9.3 mm dorsal to
area centralis and 1.75 mm nasal to the decussation line. F. Video
image and drawing of an off-beta cell (see fig. 12) viewed on the monitor
using video-overlay. The drawing of the cell has been offset slightly
above the black and white image of the HRP-labeled cell. It is possible to
plot large fields of cells at high magnification using this system. A,
B, D, and E are printed at x530. C at x1325
and F at x800 (x1,600 on the monitor).
Unsuspected dendritic differences between on- and off-center beta cells are conserved. The dendritic morphologies of on- and off-center beta cells in the wildcat differ consistently, both in the depth of their arborization (the standard criterion) and also in the shape of their proximal dendritic arbors (Fig. 11, 12). On-center cells almost invariably have 3 to 5 thin tapering and radiating primary dendrites (Figs. 11B,E, and 12) that spread out directly from the cell body and arborize neatly in the inner half of the inner plexiform layer at a distance of 6 to 7 µm from the equator of the cell body. In contrast, off-center cells almost invariably have 1 or 2 thick primary dendrites that ascend sharply, then arborize in the outer third of the inner plexiform layer (Figs. 11A, D, F and 12). Off-center arbors also have a more complex appearance than those of on-center beta cells, and the branch points of off-cell arbors are often flared outward (Fig. 11). Off-center beta cell dendrites are almost invariably more curved (spray-like) than those of on-center cells and are broader and more heavily labeled with HRP. Off-center arbors are also most commonly disposed asymmetrically with respect to the cell body, whereas on-center cell dendrites radiate symmetrically. In wildcat S1 on- and off-center cells can be reliably identified using these characteristics, without examining the depth of the dendritic arbor.
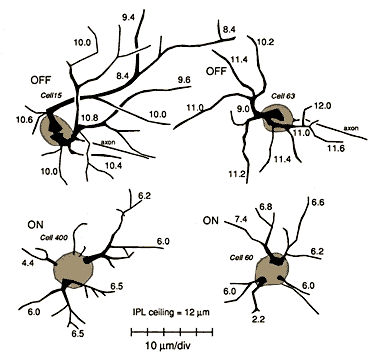
Figure 12. Typical morphology and dendritic depth of – and
off–center beta cells in the inner plexiform layer of wildcat S1. The
depths of different dendritic segments were measured from the equator of
the cell body. The interface of inner plexiform and inner nuclear layers
was typically 13 ± 1 µm from the middle of the ganglion cell layer in the
mid-periphery of this retina. There is no appreciable overlap in the depth
of secondary and tertiary dendritic branches of on– and off–center beta
cells. A video-overlay image of the off-center beta cell in the upper left
quadrant is shown in Fig. 11F.
These types of dendritic differences between on- and off-center beta cells
have not been reported previously in the domestic cat, and because these
cells have been studied in great detail, we initially assumed that this
distinction must be unique to the wildcat. However, careful reexamination
of retinas from several domestic cats revealed precisely the same
dendritic differences first noted in wildcat S1.A final difference between
off-center and on-center beta cells in both species is the size of their
somata. In wildcat S1 the volume of off-center cell bodies in dorsal
retina averaged 5300 ± 120 µm3, whereas the
volume of on-center beta cells averaged 7000 ± 160 µm3.
The difference in the domestic cat was somewhat less marked—6300 ± 170 µm3
for off-center versus 6800 ± 180 µm3 for
on-center cells. Differences between the sizes of off-center and on-center
alpha cells were not detected.
Analysis of the Optic Nerve
Marked species differences in numbers of optic axons in adults. The cross-sectional areas of the wildcats’ optic nerves were substantially greater than those of domestic cats: 2.95 mm2 in S1 and 3.24 mm2 in S2. This compares to a mean of about 2.0 mm2 in domestic cats (Williams et al., 1986). The packing density of fibers, however, is in the same range—8.8/100 µm2 in S1, 7.22/100 µm2 in S2. (Fig. 13A) and ~8.0/100 µm2 in domestic cats (Williams et al., 1983). Given these findings it is not surprising that the total population of axons is much higher in wildcats than in domestic cats: 260,000 ± 6,300 in S1 and 234,000 ± 4,500 in S2 versus 150,000 to 165,000 in domestic cats (Chalupa et al., 1984; Williams et al., 1986). The quality of fixation of the wildcat optic nerves, while more than adequate for counting axons, was not good enough to allow us to obtain reliable data on the distribution of fiber diameter. This difference between species is significant (t = 6.7, p < 0.05, two-tailed test; domestic cat data taken from Chalupa et al., 1984; Williams et al., 1986).
Table 2. Comparison of Fetal Wildcat and Domestic Cat
|
|
||
| Domestic | ||
| Fetal | Cats | |
| Trait | Wildcat | E38/E39 |
|
|
||
| Crown-rump length | 65 mm | 66–71 mm |
| Weight | 19.4 gm | 20–22 gm |
| Brain weight | 570 mg | 560 mg |
| Eye weight | 40 mg | 35–48 mg |
| Area of retinas | 30 mm2 | 20–25 mm2 |
|
|
||
Axon populations in the nerves of the fetal wildcat. Gestation in
the wildcat is of the same duration as that of the domestic cat (63 ± 3
days), weight at birth is about the same, as is the age at eye opening (Lindemann
and Rieck, 1953; Hemmer, 1976; Rodríguez de la Fuente, 1979). We therefore
estimated the age of the fetal wildcat using data on the tempo of
development in the domestic cat (Williams and Chalupa,
1982; Shatz,
1983, Williams et al.,
1986). On the basis of parameters such as crown-to-rump length, body
weight, eye mass, and retinal surface area, we conclude that the
developmental stage of this wildcat fetus corresponds very closely to that
of fetal domestic cats between embryonic day E38 and E40 (Table 2).
![]()
Figure 13. Electron micrographs of optic nerve fibers in wildcat.
A. Micrograph of a field in the left nerve of S2 at the same
magnification (x11,000) used to count axons. Due to substantial fiber and
myelin distortion, fiber diameter was not measured. B. Fibers in
the left optic nerve of a fetal wildcat. This micrograph is also
reproduced at the magnification at which the analysis was performed
(x15,000). Arrowheads mark 4 growth cones and their shanks and, in
the lower left, 1 large necrotic fiber. Comparable micrographs from a
domestic cat fetus are reproduced in figure 21 of Williams et al. (1986).
Scale bar in B represents 1 µm.
Table 3. Analysis of Optic Axons in Fetal Wildcat and Domestic Cats
|
|
||||
| Fetal wildcat | Fetal wildcat | Domestic | Domestic* | |
| Trait | Left Nerve | Right Nerve | E39 | E39 |
|
|
||||
| Axon population | 536,000±29,000 | 668,000±29,000 | 557,000±28,000 | 698,000±20,000 |
| Area of nerve (µm2) | 75,000 | 73,000 | 74,400 | 95,700 |
| Axon density (100 µm2) | 714 | 911 | 749 | 729 |
| Necrotic fibers (%) | 0.07 | 0.16 | 0.21 | 0.26 |
| Growth cones (%) | 0.17 | 0.15 | 0.20 | 0.20 |
|
|
||||
*Data from E39 domestic littermates from Williams et al. 1983,
their Table 1.
The density of axons in the fetal wildcat nerves is extremely high—an
average of 800/100 µm2 (Fig. 13B). This is very
close to the value of 735/100 µm2 in the
domestic cat fetus at E39. The right and left optic nerves of the wildcat
fetus contain 670,000 ± 30,000 and 540,000 ± 30,000 axons, respectively.
The difference between left and right nerves is high and could be due to
undercounting in the left nerve. However, there was no particular
technical difficulty in counting either nerve and it seems as likely that
the difference reflects small variation in the tempo of development in the
two eyes (see Williams et al., 1991). In any case, the estimates from this
animal are remarkably close to those of two E39 domestic cat littermates
we have previously studied—698,000 ± 20,000 and 557,000 ± 28,000 (Williams
et al., 1986). The percentages of growth cones and necrotic fibers in the
wildcat fetal nerves are also very close to those found in domestic cats
at E39 (Table 3). These two parameters are sensitive indicators of the
rate of change in the fiber population during development. The low density
of both growth cones and necrotic fibers (Table 3) indicates that the
population of fibers counted in these fetal wildcat nerves is at or very
close to the ontogenetic peak.
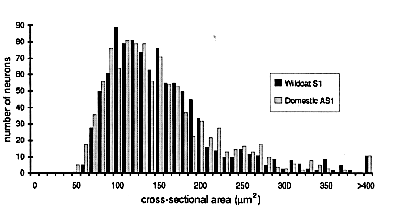
Figure 14. Size distribution of neuronal cell bodies in the LGN of
wildcat S1, and a domestic cat. In both cases, 1000 cells were measured in
sample regions extending through all laminae. Celloidin-embedded tissue
was used for this analysis. These areal measurements of cell size greatly
underestimate the in vivo values due to tissue shrinkage (Weber and Kalil,
1983).
Analysis of the Dorsal Lateral Geniculate Nucleus
The volume of the LGN differs greatly. The volume of the LGN
calculated from both frozen and celloidin sections is substantially
greater in the wildcat than in the domestic cat (Table 4). For example,
the left LGN volume of domestic cat D1 is 28.1 mm3,
whereas corresponding values for the two series of frozen sections from
the wildcats are 38.1 and 35.5 mm3. After
embedding the other hemisphere of the same domestic cat in celloidin, the
LGN volume is reduced by processing to merely 10.4 mm3.
Corresponding LGN volumes for the two wildcat hemispheres fixed, embedded,
and cut in precisely the same manner are 17.9 and 18.9 mm3.
A reasonable estimate based on our material and that kindly lent us by
Drs. Frost and Elberger (Table 4) is that the wildcat LGN has a volume
that is roughly half again as large as that of the domestic cat LGN. The
relative volumes of major components of the LGN are similar in domestic
cats and wildcats (Table 4). In both species, the A layer occupies about
37% of the nucleus, whereas the A1 layer occupies 28%.
Table 4. LGN Volume and Neuron Populations
|
|
||||||
| Case and side | Method* | Volume (mm3) | Layer A** | A1 | C-C3 | Neuron number ± SEM |
|
|
||||||
| Wildcat S1 Right | C | 18.9 | 7.0 (37%) | 5.2 (28%) | 4.5 (24%) | 774,300 ±53,000 |
| Wildcat S1 Left | F | 38.1 | 751,800±33,000 | |||
| Wildcat S2 Right | C | 17.9 | 739,700±132,000 | |||
| Wildcat S2 Left | F | 35.5 | 769,700±40,000 | |||
| Domestic D1 Left | F | 28.1 | ||||
| Domestic D1 Right | C | 10.4 | 4.0 (38) | 3.3 (32) | 2.5 (24) | 540,900±28,000 |
| Domestic Juan | C | 16.0 | 5.9 (37) | 3.9 (24) | 3.8 (24) | 475,200±35,000 |
| Domestic Storm | C | 15.6 | 5.1 (33) | 4.5 (29) | 4.0 (26) | 456,000±75,000 |
| Domestic Palomino | C | 21.7 | 514,200±62,000 | |||
| Domestic MC1 | F | 24.7 | 527,200±44,000 | |||
|
|
||||||
* C = celloidin embedded material; F = material cut frozen
**The percentage contribution of each layer to the total LGN volume is
provided in parentheses.
Marked species differences in total neuron number. As the foregoing analysis might lead one to suspect, the neuron population of the wildcat LGN is much higher than that of the domestic LGN (Table 4, right column). The populations of LGN neurons in the two wildcats were 766,000 and 754,700 (averages of left and right nuclei for S1 and S2, respectively). In comparison, the average population in five domestic cats was only 510,000 ± 26,000. These estimates include interneurons and principal neurons, and our values for the domestic cat are extremely close to those calculated by Madarász et al. (1978, their estimate: 555,000 neurons). If we make the reasonable assumption that LGN cell populations are distributed normally in both species, then this species difference is highly significant (t = 15.34, p < 0.05, two-tailed test).
Cell size in the LGN is the same. Neurons in the two species are
very closely matched in size. Sets of between 75 and 200 neurons were
measured in layers A, A1, and the C layers, in two to three sections from
each of six animals. No difference was found in cell size between domestic
cats and the wildcats (Table 5). In both species the smallest interneurons
range in size from 50 to 80 µm2, whereas
principal neurons range from 90 µm2to more than
400 µm2. Even a comparison of the size
distribution in wildcat and domestic cat cases with large differences in
LGN volume (18.9 mm3 versus 10.4 µm3)
demonstrates a remarkable similarity in the sizes of neurons in the two
species (Fig. 14).
Table 5. LGN Neuron Area (µm2)
|
|
|||
| Case | Layer A | A1 | C-C3 |
|
|
|||
| Wildcat S1 | 68 | 170 | 126 |
| Wildcat S2 | 134 | 143 | 131 |
| Domestic D1 | 166 | 160 | 103 |
| Domestic Palomino | 164 | 170 | 144 |
| Domestic Juan | 174 | 184 | 164 |
| Domestic MC1 | 153 | 159 | 100 |
|
|
|||
Note to Table 5: Cross-sectional areas (µm2)
were measured at the focal plane in which this value reach a maximum. The
standard error of the mean for each value is between 3 and 9 µm2.
All material, except that from case MC1, was embedded in celloidin. In
caseMC1, the brain was cut frozen. There is a surprising degree of
variation among the mean size of cells in the C layers of domestic cats
(range from 103 to 164). This variation appears to be independent of cell
size in the magnocellular layers.
In comparison with the domestic cat, it does not appear that any particular layer differs either in volume or in cell number more or less than the nucleus as a whole (Table 4). It is of course possible that the relative abundance of classes of LGN neurons—particularly classes 1 and 2 of Guillery (1966)—differ between wildcat and domestic cat. Unfortunately, these cell classes cannot be distinguished as easily in the LGN as they can in the retina. Estimates of cell number show much less variation than do those of the LGN volume. This is primarily because the three-dimensional counting method we have used is insensitive to differential shrinkage and does not require the application of often dubious correction factors (Williams and Rakic, 1988b).
Discussion
[Synopsis. From a quantitative standpoint, the visual systems of the Spanish wildcat and the domestic cat differ substantially. Populations of retinal ganglion cells and geniculate neurons are both 30–35% lower in the domestic cat than in the wildcat. The difference in ganglion cell populations is not matched by any change in retinal surface area, perhaps because ganglion cells make up such a small percentage of the total retinal cell population. In contrast, the LGN is smaller in the domestic cat than in the wildcat, almost precisely in proportion to the difference in total neuron number. Sizes of neurons do not differ in either part of the visual system in these closely related species.]
The descent of the domestic cat. The species Felis silvestris dates back to the middle Pleistocene (Holsteinian interglacial period; Kurtén; 1965b; 1971). During the late Pleistocene, the ancestral population of large wildcats gave way to the smaller European and North African wildcat subspecies, Felis silvestris silvestris and Felis silvestris lybica. These subspecies provided the stock from which domestic cats trace their descent over 3,000 years ago (Zeuner, 1963; Wayne et al., 1989; Fig. 15). Although details of this phylogenetic reconstruction may be modified as more material is recovered, there can be little doubt that among extant wildcats, the Spanish subspecies is morphologically closest to the Pleistocene stem population.
Dwarfing. There have been at least two distinct episodes of selection in the lineage that has led to the domestic cat. The first episode has been associated with a rapid decline in body size. This reduction occurred at the close of the Pleistocene, long before domestication (Kurtén, 1965a,b; Table 1). Such dwarfing is by no means exceptional; extremely rapid reduction in body size has been widespread among the Pleistocene mammalian fauna of Eurasia, North America, and Australia (Kurtén, 1959; Marschall and Corrunccini, 1978, Stanley, 1979, Raff and Kaufman, 1983). Rates of dwarfing have typically ranged between 4 and 40 darwins. With the exception of the Spanish wildcat subspecies, the reduction in body size in the Felis silvestris lineage has proceeded at a particularly rapid rate—about 30 darwins over the last 20,000 years (Kurtén, 1958, 1959; 1965b, his Fig. 10; one darwin is equivalent to an e-fold change over 1 million years). On the basis of our data, and conforming to allometric tendencies, the tempo of change in the visual system appears to have proceeded at a reduced, but still very rapid rate (about 15 darwins). As in other lineages, the reduction may have been linked with postglacial climatic change and human competition (Kurtén, 1965a, 1988; Van Valen, 1969; Martin and Klein, 1984).
Domestication. The second episode of intense selection has been associated with domestication. This process has undoubtedly caused changes in brain, behavior, and reproduction (Darwin, 1890; Zeuner, 1963; Stephan, 1951; Bekoff et al., 1984). However, the effects of domestication on brain structure have not yet been studied in detail (but see the exceptional work of Menner, 1939; Rohrs, 1955; and Ebinger and Lohmer, 1987). One problem facing this type of analysis is that changes in brain structure associated with altered body size must be dissociated from those effects due only to domestication. In our cellular analysis of retina and LGN of cats we have also been unable to dissociate these effects. Although we have shown that the difference in average body size of the Spanish wildcat subspecies and the domestic cat is sufficient to account for the overall reduction in brain mass in the domestic lineage (see Results), it is probable that specific selective pressures, particularly those associated with cohabitation with humans, have also generated specific changes in brain structure. A comprehensive comparative analysis of other components of the CNS and of different wildcat subspecies would be particularly useful in sorting out the relative importance of size reduction and domestication in driving brain evolution. It would be especially interesting to study the brain of the smaller North African wildcat, Felis silvestris lybica.

One Big Cat; Alexander G. Williams (age 6, June 1988).
Commissioned by K. Graehl and R. Williams. Pencil on paper.
Brain to Body Size Scaling: A Cellular Analysis of Allometry
In this study we have focused on quantitative evolutionary changes
within a small part of the central nervous system. Our data provide a
cellular perspective on allometric change in the size of the brain and
body. This cellular level of analysis complements previous work on
brain-body allometry, which has generally focused on such global factors
as ecological niche, taxonomic level, metabolic load, and developmental
timing (Eisenberg and Wilson, 1978; Lande, 1979; Armstrong, 1982; Martin
and Harvey, 1985, assessed in Mann et al., 1988; Preuss, 1992).
The average size difference between the Spanish wildcat and
domestic cat is approximately twofold—6 to 7 kg for the wildcat versus 2.5
to 3.5 kg for the domestic cat. The 25 to 30% difference in average brain
weight—31 to 37 gm for the wildcats versus 25 to 29 gm for the domestic
cat—is almost precisely what one would predict from the allometric
relationship calculated for felids (Davis, 1962). Data on wildcat brain
and body weights from several sources [summarized by Röhrs (1955)] also
affirm that differences in brain weights of other wildcat subspecies and
domestic cat conform closely to allometric predictions. We note that the
two wildcats we studied weighed less than is typical, presumably due to
their prolonged captivity. However this does not compromise our
observation that their brains are much larger than those of domestic cats,
and that the weights of their brains are consistent with allometric
predictions based on the average body weight of the Spanish wildcat.
We can now ask and answer more interesting questions: how has the
allometric reduction in brain weight in the cat lineage been accomplished?
What are the cellular correlates or mechanisms? Our results reveal that in
the primary visual system, the decrease in brain mass has been brought
about strictly by reductions in cell number. In particular, we have shown
that populations of retinal ganglion cells and neurons in the LGN are
reduced 30 to 35%. This reduction matches the reduction in total brain
weight. There is no evidence for any change in neuron size or in the
amount of neuropil. If one is willing to generalize our findings in retina
and the LGN to the entire CNS, then it follows that the total population
of neurons is about 30% less in domestic cats than in Spanish wildcats.
These data suggest that rapid evolutionary modulation in brain size
is brought about primarily by changes in cell number. Wikler et al. (1989)
have also noted that the evolution of the retina in the Cricetidae family
has primarily involved change in cell number. In contrast, long-term brain
evolution encompassing many millions of years of divergence has often been
associated with substantial changes in cell type and cell size, dendritic
and electrophysiological properties, as well as the number of
cytoarchitectonic areas (Holloway, 1968; Purves et al., 1986; Kaas 1987;
Purves, 1988; Bekker and Stevens, 1990; Reiner, 1991). Such changes must
usually involve more far-reaching genetic and epigenetic reprogramming
than those associated with fluctuations in cell number alone.
Allometric exceptions. We found several interesting exceptions
in which numbers of cells appear not to have changed as one would predict
from the allometric relationship between brain and body size. First, the
population of rods in the domestic cat equals, and may even exceed, that
in the wildcat. Rod densities in the area centralis and along part of the
horizontal axis in domestic cats (our data and Steinberg et al., 1973) are
higher than those measured in the male wildcat S1. Although it would be
unwise to read too much into this difference, given the variability in rod
and cone densities in other species (Curcio et al., 1990; Wikler et al.,
1990; Williams, 1991), we can certainly say that the rod population is as
high (and possibly higher) in the domestic cat as in the Spanish wildcat.
Similarly, the population of alpha ganglion cells in the domestic cat has
not undergone any reduction. In fact, densities of alpha cells at the
center of the area centralis may be slightly higher in domestic cats than
in the wildcat retinas that we studied. This evolutionary stasis in alpha
cell number stands in marked contrast to the sharp decline in the total
ganglion cell population.
These two intriguing exceptions demonstrate that allometric changes
in brain mass represent averages that can mask substantial variation in
the magnitude of change among different neuron subpopulations (Harvey and
Krebs, 1990). As we and others have shown, these differential effects on
neuron populations may be prominent even when the evolutionary and genetic
separation between two species, or even two strains, is exceedingly small
(Smith, 1928; Holloway, 1968; Wimer et al., 1976; Ebinger and Lohmer,
1987). In the domestic cat lineage, selective pressures appear to have
counterbalanced allometric tendencies and thereby have maintained scotopic
and motion sensitivity by leaving intact genetic and epigenetic mechanisms
responsible for generating and maintaining populations of rods and alpha
ganglion cells. In contrast, populations of cones and beta ganglion cells
(particularly in the area centralis) have not been spared a sharp decline.
Cellular selectivity of this sort serves to emphasize the underlying
genetic and epigenetic complexity of allometric relations.
A Subtle Difference Between On- and Off-Center Beta Cell Dendrites is Conserved in Both Felis Species
We initially thought that the marked dendritic differences noted between on- and off-center beta cells (symmetric versus asymmetric and thick versus thin primary dendrites) were unique to the wildcat (Figs. 11 and 12)—that we had finally detected a significant qualitative difference. However, careful inspection of domestic cat retinas revealed precisely the same structural differences between on- and off-center beta cells. Although we are the first to comment on this dichotomy between on- and off-center beta cells, some of these differences can be discerned in figures of previous papers on domestic cat ganglion cells (e.g., Wässle et al., 1981a; Rowe and Dreher, 1982; Stanford, 1987). These marked structural differences in dendrites suggest that a functional reanalysis such as that of Koch et al. (1982) would be revealing, and that these two subtypes of neurons may have unsuspected electrotonic differences. From an evolutionary perspective, it is also noteworthy that relatively subtle, previously undetected, dendritic differences have been conserved in this lineage despite large differences in numbers of these cells. An analysis of on- and off-center cells in other mammals would be of interest to assess the taxonomic breadth of this dichotomy.
Developmental Mechanisms and Brain Evolution
An intriguing question raised by these findings is at what stage of
development and in what manner the great differences between the brains of
wild and domestic species arise. As mentioned in the Results section, the
length of gestation in wild and domestic cats is about the same (63 days),
birth weight is roughly the same (100 ± 20 gm), and eye opening occurs at
the same age (7 to 10 days after birth). One explanation for the reduction
in the size of the domestic cat’s brain is that far fewer neurons are
generated early in development. A more counterintuitive, but equally
possible alternative, is that the same numbers of neurons are generated in
the domestic cat as in the wildcat, but that many more of these cells are
subsequently eliminated. For reasons listed below, we think that the
decrease in brain size in the domestic lineage is probably associated with
an increase in the incidence of cell death.
Cell death in the domestic cat retina is particularly severe—80% of
all ganglion cells die in prenatal and early postnatal development
(Williams et al., 1986). This magnitude of cell death exceeds by a wide
margin that in other vertebrates in which ganglion cell death has yet been
studied, including rhesus macaque and human (Rakic and Riley, 1983; Provis
et al., 1985). As suggested in previous work (Williams et al., 1986), the
excessive production of ganglion cells in the domestic cat fetus may be an
ontogenetic trait held over from its large wildcat ancestor—a form of
phylogenetic inertia (Harvey and Purvis, 1991). The elimination of this
surplus may correspond to what Glücksmann (1951) has referred to as
phylogenetic cell death. Our remarkable, but admittedly tentative finding
that the numbers of optic axons in wildcat and domestic cat fetuses do not
differ appreciably early in development is consistent with a role for
ganglion cell death operating at a later stage of development in
generating the marked species differences seen at maturity.
The idea that brain evolution may be associated with late fetal and
early neonatal modulation in the incidence of cell death is also
consistent with an observation first made by von Baer (1828) that early
stages of development in related species tend to be relatively uniform
whereas late stages tend to differ (Gould, 1977; Buss, 1987). A
developmental and evolutionary shift in the onset or duration of cell
death in a lineage may be just as effective in changing CNS structure as a
heterochronic shift in the onset or duration of cell proliferation (Wikler
and Finlay, 1989; Goldowitz, 1989). Of course, the modulation of cell
death is ultimately limited by the degree of neuron overproduction.
Consequently this mechanism cannot provide an explanation for the
long-term phyletic increase in brain size that is prominent in some
mammalian lineages. For instance, it is improbable that the brain of Homo
sapiens is larger than that of Australopithecine ancestors simply because
fewer neurons are lost at a late stage of development.
Our argument implies that the severity of cell death is generally
elevated in the CNS of domestic cats in comparison to that in the CNS of
Spanish wildcats—a testable prediction. It may be the case that large
litter size and the high metabolic load of late pregnancy on smaller
domestic females serve as a biochemical brake that eliminates many neurons
when they first become active. This metabolic interpretation provides some
rationale for the loss of normally connected neurons (Williams and Herrup,
1988; Chalupa and Lia, 1991; Oppenheim, 1991).
The modulation of rates of cell death rather than of rates of cell
production could be of selective advantage in making possible rapid and
well-matched evolutionary changes in the size of interconnected neuron
populations (Katz and Lasek, 1978; Finlay et al., 1987). Seen in this
light, the overproduction of neurons in a species represents a concealed
source of variation that can be easily exploited in response to selective
pressure by a simple change in the incidence of cell death (Williams and
Herrup, 1988). There is a great deal of evidence that the modulation of
cell death is in large part governed by highly flexible trophic feedback
mechanisms (Purves, 1988). Such responsive and contingent mechanisms may
effectively loosen the grip of epigenetic constraints on brain evolution
(Katz and Lasek, 1978; Hamburger and Oppenheim, 1982). These ideas are
compatible with the neatly matched 30% difference in both retinal ganglion
cell and LGN neuron populations that we have documented in this study.
Modes of development are subject to selection both at the level of
the individual and at the level of the species. The presence or absence
within a lineage of a mechanism involving the overproduction and
subsequent loss of neurons via trophic interactions may provide a concrete
developmental basis for differences in rates of speciation (Purves, 1988,
p. 174) and consequently, for selection at, or even above, the species
level (Stanley, 1975; Vrba, 1983; Raff and Kaufman, 1983).
Acknowledgements
This research was supported in part by grant DGICYT PB88-0170 and a grant from the NIH. We are especially indebted to Mariano Sanz Pech and Antonio Pérez Alonso-Geta of the Institute for the Conservation of Nature, Madrid, Spain, for wildcat material. Rosa Sánchez Lozano provided expert technical assistance in processing celloidin-embedded material. We thank Drs. Leo Chalupa, Andrea Elberger, and Douglas Frost, for the generous use of material from their collections of domestic cat tissue, and Drs. Leo Chalupa, Robert Foehring, Dan Goldowitz, Todd Preuss, Anton Reiner, and Michael Rowe for critical comments on drafts of this paper. We thank Alexander G. Williams for production and editing of the HTML edition of this paper.

|
Close enough... |
References
Armstrong E (1982) Relative brain size and metabolism in mammals. Science 220:1302–1304.
Bekker JM, Stevens CF (1990) Two different ways evolution makes neurons larger. Prog Brain Res 83:37–45.
Benveniste RE (1985) The contributions of retroviruses to the study of mammalian evolution. In: Molecular evolutionary genetics (MacIntyre RJ, ed), pp 359–417. New York: Plenum.
Boycott BB, Wässle H (1974) The morphological types of ganglion cells of the domestic cat’s retina. J Physiol (Lond) 240:397–419.
Brændgaard H, Gundersen HJG (1986) The impact of recent stereological advances on quantitative studies of the nervous system. J Neurosci Meth 18:39–78.
Buss LW (1987) The evolution of individuality. Princeton: Princeton Univ Press.
Carroll RL (1988) Vertebrate paleontology and evolution. New York: Freeman.
Chalupa LM, Williams RW, Henderson Z (1984) Binocular interaction in the fetal cat regulates the size of the ganglion cell population. Neuroscience 12:1139–1146.
Chalupa LM, Lia B (1991) The nasotemporal division of retinal ganglion cells with crossed and uncrossed projections in the fetal rhesus monkey. J Neurosci 11:191–202.
CITES (1991) Convention on international trade in endangered species of wild flora and fauna. Appendices I–III. Endangered species convention. Oct 1, 1991, 50 CFR 23.23. Endangered and threatened wildlife and plants. July 15, 1991:50 CFR 17.11.
Collier G, O’Brien SJ (1985) A molecular phylogeny of the Felidae: immunological distance. Evolution 39:473–487.
Cooper ML, Pettigrew JD (1979) The decussation of the retinothalamic pathway in the cat, with a note on the major meridians of the cat’s eye. J Comp Neurol 187:285–312.
Clutton–Brock J (1981) Domesticated animals from early times. Cambridge: Cambridge Univ Press.
Curcio CA, Sloan KR, Kalina RE, Hendrickson AE (1990) Human photoreceptor topography. J Comp Neurol 292:497–523.
Darwin C (1890) The variation of animals and plants under domestication. 2nd ed, revised. New York: Appleton and Co.
Davis DD (1962) Allometric relationships in lions vs. domestic cats. Evolution 16:505–514.
Ebinger P, Lohmer R (1987) A volumetric comparison of brains between greylag geese (Anser anser L.) and domestic geese. J Hirnforsch 28:291–299.
Edinger T (1948) Evolution of the horse brain. Geol Soc Amer Mem 25:1–177.
Edinger T (1966) Brains from 40 million years of Camelid history. In: Evolution of the forebrain (Hassler R, Stephan H, eds), pp 153–161. Stuttgart: Thieme.
Eisenberg JF (1981) The mammalian radiation. An analysis of trends in evolution, adaptation, and behavior. Chicago: Univ Chicago Press.
Eisenberg JF, Wilson DE (1978) Relative brain size and feeding strategies in the Chiroptera. Evolution 32:740–751.
Eldredge N, Gould SJ (1972) Punctuated equilibria: an alternative to phyletic gradualism. In: Models in Paleobiology. (Schopf TJM, ed), pp 82–115. San Francisco: Freeman, Cooper, and Co.
Finlay BL, Wikler KC, Sengelaub DR (1987) Regressive events in brain development and scenarios for vertebrate brain evolution. Brain Behav Evol 30:102–117.
Glücksmann A (1951) Cell deaths in normal vertebrate ontogeny. Biol Rev 26:59–86.
Goldowitz D (1989) Cell allocation in mammalian CNS formation: Evidence from murine interspecies aggregation chimeras. Neuron 3:705–713.
Gould SJ (1977) Ontogeny and phylogeny. Cambridge, Mass: Harvard Univ Press.
Gould SJ, Eldredge N (1986) Punctuated equilibrium at the third stage. Syst Zool 35:143–148.
Guillery RW (1966) A study of Golgi preparations from the dorsal lateral geniculate nucleus of the adult cat. J Comp Neurol 128:21–50.
Haltenorth T (1953) Die Wildkatzen der Alten Welt. Eine Ü;bersicht über die Untergattung Felis. Leipzig: Akademische Verlagsgesellschaft, Geest and Portig.
Hamburger V, Oppenheim RW (1982) Naturally occurring neuronal death in vertebrates. Neurosci Comm 1:39–55.
Harvey PH, Krebs JR (1990) Comparing brains. Science 249:139–146.
Harvey PH, Purvis A (1991) Comparative methods for explaining adaptations. Nature 351:619–624.
Hemmer H (1976) Gestation period and postnatal development in Felids. In: The world’s cats, Vol 3, No 2, (Eaton RL, ed) pp 90–100. Seattle: Carnivore Research Institute, Burke Museum, Univ Washington.
Holloway RL (1968) The evolution of the primate brain: some aspects of quantitative relations. Brain Res 7:121–172.
Hughes A (1975) A quantitative analysis of the cat retinal ganglion cell topography. J Comp Neurol 163:107–128.
Jacobs DS, Perry VH, Hawken MJ (1984) The postnatal reduction of the uncrossed projection from the nasal retina in the cat. J Neurosci 4:2425–2433.
Jerison HJ (1973) Evolution of the brain and intelligence. New York: Academic Press.
Jerison H (1979) Brain, body and encephalization in early primates. J Hum Evol 8:615–635.
Kaas JH (1987) The organization and evolution of neocortex. In: Higher brain function: recent explorations of the brain’s emergent properties (Wise SP, ed), pp 347–378. New York: Wiley.
Katz MJ, Lasek RJ (1978) Evolution of the nervous system: Role of ontogenetic mechanisms in the evolution of matching populations. Proc Natl Acad Sci USA 75:1349–1352.
Kirby MA, Chalupa LM (1986) Retinal crowding alters the morphology of alpha ganglion cells. J Comp Neurol 251:532–541.
Koch C, Poggio T, Torre V (1982) Retinal ganglion cells: a functional interpretation of dendritic morphology. Phil Trans R Soc Lond B 298:227–264.
Kurtén B (1954) Observations on allometry in mammalian dentition; Its interpretation and evolutionary significance. Acta Zool Fennica 76:1–122.
Kurtén B (1958) A differentiation index, and a new measure of evolutionary rates. Evolution 12:146–157.
Kurtén B (1959) Rates of evolution in fossil mammals. Cold Spring Harbor Symp Quant Biol 24:205–215.
Kurtén B (1965a) The Carnivora of the Palestine caves. Acta Zool Fennica 107:1–74.
Kurtén B (1965b) On the evolution of the European wild cat, Felis silvestris Schreber. Acta Zool Fennica 111:1–29.
Kurtén B (1971) The age of mammals. New York: Columbia Univ Press.
Kurtén B (1988) On evolution and fossil mammals. New York: Columbia Univ Press.
Lande R (1979) Quantitative genetic analysis of multivariate evolution applied to brain:body size allometry. Evolution 33:402–416.
Latimer HB (1938) The weights of the brain and of its parts, of the spinal cord and of the eyeballs in the adult cat. J Comp Neurol 68:395–404.
Lia B, Williams RW, Chalupa LM (1986) Does axonal branching contribute to the overproduction of optic nerve fibers during early development of the cat’s visual system. Dev Brain Res 25:296–301.
Lindemann W, Rieck W (1953) Beobachtungen bei der Aufzucht von Wildkatzen. Zschrft f Tierpsychol 10:92–119.
Madarász M, Gerle J, Hajdu F, Somogyi G, Tömböl T (1978) Quantitative histological studies on the lateral geniculate nucleus in the cat. II. Cell numbers and densities in several layers. J Hirnforsch 19:159–164.
Mann MD, Glickman SE, Towe AL (1988) Brain/body relations among myomorph rodents. Brain Behav Evol 31:111–124.
Marschall LG, Corrunccini RS (1978) Variability, evolutionary rates, and allometry in dwarfing lineages. Paleobiology 4:101–119.
Martin PS, Klein RG (eds) (1984) Quaternary extinctions: a prehistoric revolution. Tucson: Univ Arizona Press.
Martin RD, Harvey PH (1985) Brain size allometry. Ontogeny and phylogeny. In: Size and scaling in primate biology (Jungers WL, ed), pp 147–173. New York: Plenum.
Menner E (1939) Vergleichende Untersuchungen über die Retina wildlebender und domestizierter Caniden. Z Naturw 93:77–88.
Mountcastle VB (1980) Medical physiology, 14th ed, St. Louis: Mosby.
Nikara T, Bishop PO, Pettigrew JD (1968) Analysis of retinal correspondence by studying receptive fields of binocular single units in the cat striate cortex. Exp Brain Res 6:353–372.
Oppenheim RW (1991) Cell death during development of the nervous system. Annu Rev Neurosci 14:453–501.
Otsuka R, Hassler R (1962) Über Aufbau und Gliederung der corticalen Sehsphare bei der Katze. Arch Psychiat Z ges Neurol 203:212–234.
Pagel MD, Harvey PH (1989) Taxonomic differences in the scaling of brain on body weight among mammals. Science 224:1589–1593.
Peichl L, Wässle H (1981) Morphological identification of on– and off–centre brisk transient (Y) cells in the cat retina. Proc R Soc Lond B 212:139–156.
Perry VH, Linden R (1982) Evidence for dendritic competition in the developing retina. Nature 297:683–685.
Preuss TM (1992) The role of neurosciences in primate evolutionary biology: Historical commentary and prospectus. In: Primates and their relatives in phylogenetic perspective (MacPhee RDE, ed), pp xx–xx. New York: Plenum.
Provis JM, van Driel D, Billson FA, Russell P (1985) Human fetal optic nerve: overproduction and elimination of retinal axons during development. J Comp Neurol 238:92–101.
Purves D (1988) Body and Brain. A Trophic Theory of Neural Connections. Cambridge, Mass: Harvard Univ Press.
Purves D, Rubin E, Snider WD, Lichtman J (1986) Relation of animal size to convergence, divergence, and neuronal number in peripheral sympathetic pathways. J Neurosci 6:158–163.
Radinsky LB (1973) Evolution of the canid brain. Brain Behav Evol 11:169–202.
Radinsky LB (1975) Evolution of the felid brain. Brain Behav Evol 11:214–254.
Radinsky LB (1981) Brain evolution in extinct South American ungulates. Brain Behav Evol 18:169–187.
Raff RA, Kaufman TC (1983) Embryos, genes, and evolution. The developmental–genetic basis of evolutionary change. New York: Macmillan.
Rakic P, Riley K (1983) Overproduction and elimination of retinal axons in the fetal rhesus monkey. Science 219:1441–1444.
Reiner A (1991) A comparison of neurotransmitter–specific and neuropeptide–specific neuronal cell types present in the dorsal cortex in turtles with those present in the isocortex in mammals: Implications for the evolution of isocortex. Brain Behav Evol 38:53–91.
Reinoso–Suárez F (1961) Topographischer Hirnatlas der Katze für experimental–physiologische Untersuchungen. Darmstadt: E. Merck.
Rodr&icacute;guez de la Fuente F (1979) El gato montes. In: Fauna Iberica y Europea. Vol. VI: Bosque Caducifolio. Bosque de Coníferas y Taiga. pp 60–69. Barcelona: Salvat Editores.
Röhrs M (1955) Vergleichende Untersuchungen an Wild– und Hauskatzen. Zool Anz 155:53–69.
Rosen GD, Harry JD (1990) Brain volume estimation from serial section measurements: a comparison of methodologies. J Neurosci Meth 35:115–124.
Rowe MH, Stone J (1976) Properties of ganglion cells in the visual streak of the cat’s retina. J Comp Neurol 167:99–126.
Rowe MH, Dreher B (1982) Functional morphology of beta cells in the area centralis of the cat’s retina: a model for the evolution of central retinal specializations. Brain Behav Evol 21:1–23.
Schindewolf OH (1950) Grundfragen der Palaontologie. Stuttgart: Schwarzerbart.
Shatz CJ (1983) The prenatal development of the cat’s retinogeniculate pathway. J Neurosci 3:482–499.
Smith LA (1928) A comparison of the number of nerve cells in the olfactory bulbs of domesticated albino and wild Norway rats. J Comp Neurol 45:483–499.
Stanford LR (1987) X–cells in the cat retina: relationships between the morphology and physiology of a class of cat retinal ganglion cells. J Neurophysiol 58:940–964.
Stanley SM (1975) A theory of evolution above the species level. Proc Natl Acad Sci USA 72:646–650.
Stanley SM (1979) Macroevolution. Pattern and process. San Francisco: Freeman.
Steinberg RH, Reid M, Lacy PL (1973) The distribution of rods and cones in the retina of the cat (Felis domesticus). J Comp Neurol 148:229–248.
Stone J (1978) The number and distribution of ganglion cells in the cat’s retina. J Comp Neurol 180:753–772.
Stone J, Rapaport DH, Williams RW, Chalupa LM (1982) Uniformity of cell distribution in the ganglion cell layer of prenatal cat retina: Implications for mechanisms of retinal development. Dev Brain Res 2:231–242.
Van Valen L (1969) Evolution of communities and late Pleistocene extinctions. North Amer Paleont Convention, Chicago Proc:469–485.
von Baer KE (1828) Über Entwickelungsgeschichte der Thiere. Beobachtung und Reflexion. Part I. Königsberg: Gebrüdern Bornträger (available at Beineke Library, Yale University).
Vrba ES (1983) Macroevolutionary trends: new perspectives on the roles of adaptation and incidental effect. Science 221:387–389.
Wässle H, Levick WR, Cleland BG (1975) The distribution of the alpha type of ganglion cells in the cat’s retina. J Comp Neurol 159:419–438.
Wässle H, Riemann RJ (1978) The mosaic of nerve cells in the mammalian retina. Proc R Soc Lond B 200:441–461.
Wässle H, Boycott BB, Peichl L (1981a) Morphology and mosaic of on– and off–beta cells in the cat retina and some functional considerations. Proc R Soc Lond B 212:177–195.
Wässle H, Peichl L, Boycott BB (1981b) Morphology and topography of on– and off–alpha cells in the cat retina. Proc R Soc Lond B 212:157–175.
Wayne RK, Benveniste RE, Janczewski DN, O’Brien SJ (1989) Molecular and biochemical evolution of the Carnivora. In: Carnivore behavior, ecology, and evolution. (Gittleman JL ed), pp 465–494. Ithaca New York: Cornell Univ Press.
Weber AJ, Kalil RE (1983) The percentage of interneurons in the dorsal lateral geniculate nucleus of the cat and observations on several variables that affect the sensitivity of horseradish peroxidase as a retrograde marker. J Comp Neurol 220:336–346.
Wikler KC, Finlay BL (1989) Developmental heterochrony and the evolution of species differences in retinal specializations. In: Development of the vertebrate retina. (Finlay BL, Sengelaub DR, eds), pp 227–246. New York: Plenum.
Wikler KC, Perez G, Finlay, BL (1989) Duration of retinogenesis: its relationship to retinal organization in two Crisitine rodents. J Comp Neurol 285:157–176.
Wikler KC, Williams RW, Rakic P (1990) Photoreceptor mosaic: number and distribution of rods and cones in the rhesus monkey retina. J Comp Neurol 297:499–508.
Williams RW (1991) The human retina has a cone–enriched rim. Vis Neurosci 6:403–406.
Williams RW, Chalupa LM (1982) Prenatal development of the retinocollicular projection in the cat: an anterograde transport study. J Neurosci 2:604–622.
Williams RW, Chalupa LM (1983) An analysis of axon caliber within the optic nerve of the cat: evidence of size groupings and regional organization. J Neurosci 3:1554–1564.
Williams RW, Bastiani MJ, Lia B, Chalupa LM (1986) Growth cones, dying axons, and developmental fluctuations in the fiber population of the cat’s optic nerve. J Comp Neurol 246:32–69.
Williams RW, Herrup K (1988) The control of neuron number. Annu Rev Neurosci 11:423–453.
Williams RW, Rakic P (1988a) Elimination of neurons from the rhesus monkey’s lateral geniculate nucleus during development. J Comp Neurol 272:424–436.
Williams RW, Rakic P (1988b) Three–dimensional counting: An accurate and direct method to estimate numbers of cells in sectioned material. J Comp Neurol 278:344–352.
Williams RW, Borodkin M, Rakic P (1991) Growth cone distribution patterns in the optic nerve of fetal monkeys: Implications for mechanisms of axon guidance. J Neurosci 11:1081–1094.
Wimer RE, Wimer CC, Vaughn JE, Barber RP, Balvanz BA, Chernow CR (1976) The genetic organization of neuron number in Ammon’s horns of house mouse. Brain Res 118:219–243.
Wong ROL, Hughes A (1987a) Developing neuronal populations of the cat retinal ganglion cell layer. J Comp Neurol 262:473–495.
Wong ROL, Hughes A (1987b) The morphology, number, and distribution of a large population of confirmed displaced amacrine cells in the adult cat retina. J Comp Neurol 255:159–177.
Zeuner FE (1963) A history of domesticated animals. New York: Harper and Row.
Contents
Abstract
Introduction
Materials and Methods
Comparison of body and brain weights
Analysis of retinal structure
Analysis of ganglion cell numbers
Analysis of the LGN
Evolutionary history of the domestic cat
Dwarfing
Domestication
Brain to body size scaling
ON- and OFF-center retinal ganglion cells
Development and brain evolution
Received April 14, 1992; revised July 13, 1992; accepted July
15, 1992; published January 1993.
HTML edition created by AGW and RWW Jan 1997; updated Aug. 4, 1998 by RWW.
This paper was reviewed by Sandra Blakeslee in The New York Times.
Since 11 August 98
