Elimination of Neurons from the Lateral Geniculate Nucleus of Rhesus Monkeys during Development
Robert W. Williams and Pasko Rakic
Section of Neuroanatomy, Yale University School of Medicine, 333 Cedar Street, New Haven CT 06511
The Journal of Comparative Neurology 272:424–436 (1988)
 Notes to the reader: This is a revised version of a paper published
in The Journal of Comparative Neurology in 1988. Modifications are delimited
by brackets [...].
Notes to the reader: This is a revised version of a paper published
in The Journal of Comparative Neurology in 1988. Modifications are delimited
by brackets [...].
Enlarging images: Thumbnail versions of all figures are embedded in the paper. A better image—usually under 100K—will download into a separate window if you click on the thumbnail image. If you have a large enough monitor, drag this second figures window beside the text window. Finally, high-resolution images—usually under 1 MB—that almost match the quality of the original prints can be downloaded by selecting the text at the bottom of each legend. These image files can be viewed with Adobe Photoshop, NIH Image, or equivalent.
Revised HTML edition (http://www.nervenet.org/papers/RHESUS_LGN.html) copyright © 1998 by Robert W. Williams.
|
Table of Contents
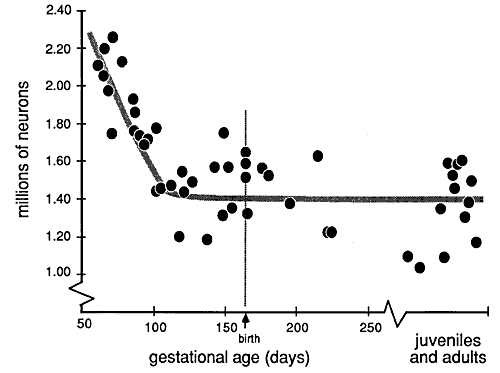
ABSTRACTThe timing, magnitude, and spatial distribution of neuron elimination was studied in the dorsal lateral geniculate nucleus of 54 rhesus monkeys (Macaca mulatta) ranging in age from the 48th day of gestation to maturity. Normal and degenerating cells were counted in Nissl-stained sections using video-enhanced differential inference contrast optics and video-overlay microscopy. Before embryonic day 48 (E48), the geniculate nucleus contains 2,200,000 ± 100,000 neurons. Eight-hundred thousand of these neurons are eliminated over a 50-day period spanning the middle third of gestation. Neurons are lost at an average rate of 300 an hour between E48 and E60, and at an average rate of 800 an hour between E60 and E100. Very few neurons are lost after E100, and as early as E105, the population has fallen to the adult average of 1,430,000 ± 90,000. Degenerating neurons are far more common in the magnocellular part of the nucleus than in the parvocellular part. In 19 of 27 cases, the number of neurons is greater on the right than on the left side. The right-left asymmetry averages 8.5% and is significant (chi-square = 17.7, p <0.02). The elimination of neurons in the nucleus begins before the depletion of retinal axons, and this raises the possibility that the number of geniculate neurons determines the final size of the retinal ganglion cell population rather than vice versa. The period of cell death precedes the emergence of cell layers in the geniculate, the establishment of geniculocortical connections, and the formation of ocular dominance columns. [Key words:neuron death, dorsal lateral geniculate nucleus, rhesus monkey, primate development]
INTRODUCTIONLittle is known about the onset, duration, magnitude, or spatial distribution of neuron elimination in the vertebrate visual system. The analysis of naturally occurring neuron loss has been difficult because embryonic neurons are small and are packed together so tightly, and because the boundaries of visual centers are indistinct at early developmental stages. In this study we have taken advantage of video-enhanced differential interference contrast optics (DIC) and video-overlay microscopy to overcome some of these problems, and using these methods we have been able to estimate the key parameters of neuron elimination in the dorsal lateral geniculate nucleus of a primate—the rhesus monkey, Macaca mulatta—from early in prenatal life through to maturity. The simplicity of the retino-geniculo-cortical pathway of macaques and its similarity to that of humans makes it an excellent system in which to study interactions that control numbers of neurons in different parts of the visual system (Rakic and Williams, 1986; Williams and Herrup, 1988). More than 90% of all retinal ganglion cells in macaques project only to the lateral geniculate nucleus and more than 90% of all geniculate neurons project only to primary visual cortex (Perry et al, 1984; Norden and Kaas, 1978; Hamori et al., 1983; Pasik et al., 1986). Furthermore, we know when neurons are generated and when synaptic contacts are established in the visual system of macaques (Rakic, 1974, 1976, 1977ab; LaVail et al., 1983; Shatz and Rakic, 1981; Hendrickson and Rakic, 1977; Kostovic and Rakic, 1984; Nishimura and Rakic, 1985, 1987; Bourgeois and Rakic, 1983). Consequently, changes in the population of neurons in the geniculate nucleus can now be studied profitably with respect to other cellular events in development—for instance, the innervation of the nucleus by retinal and cortical axons, the elimination of retinal ganglion cells and their axons, the lamination of the nucleus, the innervation of primary visual cortex by geniculate fibers, and the development of ocular dominance columns.
MATERIAL AND METHODSThis study is based on an analysis of 54 animals ranging in age from E48 to maturity. Normal monkey fetuses of known gestational age were delivered by cesarian section and while anesthetized with Halothane were perfused through the heart with saline and mixed aldehydes (Rakic, 1972). Postnatal monkeys were anesthetized with sodium pentobarbital and were perfused with either formalin or mixed aldehydes. Brains were sectioned serially in the coronal (n = 50) or horizontal (n = 4) planes, and a minimum of every tenth section was mounted and stained with cresyl violet or Toluidine blue. Much of the material was taken from a pre-existing rhesus macaque brain collection, and because several embedding and cutting protocols had been used to prepare this material, it was necessary to ensure that the estimates did not depend on large or small differences in processing. Four control cases (E61, E66, E128, and P0) were prepared in which right and left sides of the brain were embedded in different media (celloidin vs. paraffin, or celloidin vs. a 30% solution of sucrose) and cut at different thicknesses (20 and 35 µm). Right versus left differences for this group averaged 11.4% compared to an average difference of 8.9% for all other material. This difference is not significant. In addition to these control cases, 11 prenatal brains were embedded only in celloidin and sectioned at 35 µm (Yakovlev, 1970), while an additional 11 brains were cut frozen at 30 or 40 µm. Two neonatal brains (P0 and P11) were were cut in paraffin at 20 µ;m whereas one other neonatal brain (P15) was cut frozen at 40 µm. All other postnatal brains (n = 18) were embedded in celloidin and sectioned at 35 µm. Finally, 7 of the youngest cases (E48, 50, 54, 55, 59, 60, and 70) were embedded in polyester wax and sectioned at 8 µm (Rakic, 1977). These latter cases were used only to estimate the fraction of degenerating cells in the thalamus (see below). Immunocytochemistry. To corroborate criteria used to distinguish neurons from glial cells (see Recognition of neurons), we examined sections of the thalamus immunoreacted with antibodies directed against glial fibrillary acid protein (GFAP) at three ages—E72, E78, and E90. Methods and controls used to prepare tissue are described in papers by Levitt and Rakic (1980) and Levitt et al. (1983). Tissue was cut on a Vibratome at 60 µm, and sections were incubated for 15 h in a 1:500 dilution of rabbit antiserum and 5% normal goat serum. Avidin-biotin conjugate and diaminobenzidine were subsequently used to localize GFAP. Estimation of cell numbers Direct estimates of neuron number in the geniculate nucleus were obtained for 47 rhesus monkeys. In 27 of these cases, estimates were obtained from both sides, and the means of right and left sides were used in all subsequent calculations. The accuracy of the estimates depends crucially on five factors:
The procedures and problems associated with each of these factors are considered below.
Recognition of neurons. Counts were performed at a final magnification of x2500 using a x100 oil immersion objective and video-enhanced DIC optics (Inoue, 1986). The following morphological criteria were used to distinguish neurons from glial cells, endothelial cells, and pericytes:
We consider problems associated with the distinction of cells during this period, and how these problems may affect the interpretation, in the Results section. Accuracy of counting. The number of neurons in a single counting frame ranged from 5 to 250. Neurons were counted if they were completely within the margins of the frame or if they intersected only its upper or right sides (Gundersen, 1977). Even at a magnification of 2500, it is possible to miss 10% of the cells, particularly when the packing density is high. To improve the accuracy of counting we have used video-enhanced DIC optics in combination with a video-overlay system. A camera (Hamamatsu C2400) was mounted on the microscope, and the field of cells was viewed on a video monitor (Sony PVM-1271Q). The outlines of each cell (nucleus or nucleolus) were traced at x2500 on the screen of the monitor using a digitizing tablet as an input device. Because cells were marked and measured on the monitor, errors of omission and commission were easily detected and corrected. Estimation of the total volume. Outlines of the geniculate nucleus were drawn and measured at evenly spaced intervals through the entire nucleus. The length of the nucleus orthogonal to the cutting plane was estimated by multiplying the mean section thickness by the number of sections, and the total volume was calculated by multiplying the average area by the length.
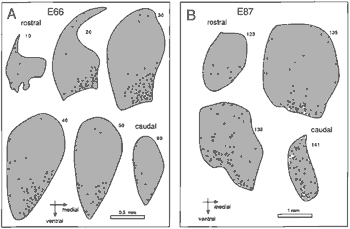
Sampling procedures. The density of neurons in the geniculate nucleus varies with position. Neurons are scattered more widely in the interlaminar zones than within the cell layers, and cell densities are lower in magnocellular layers than in parvocellular layers even as early as E86. In contrast, there are only negligible regional differences within layers. However, because the overall laminar makeup of the nucleus—that is, the combination and spacing of layers present in any given section—differs from front to back or from top to bottom (Kaas et al., 1978), and because of differences in the distribution of fibers of passage, there are rostrocaudal gradients in cell packing density. For instance, the mean difference along the rostrocaudal axis varies from 15 to 30% in postnatal animals (Kupfer et al., 1967). To get a balanced sample, all layers were examined in a series of fields extending from the ventral-most layer (layer 1 or the superficial layer S) through to layer 6 at three or more levels. The difference between our standard sampling method (one translaminar probe per section, typically three to six sections examined per nucleus) and a more extensive, uniform sampling procedure (up to 10 probes per section, 8 to 10 sections examined per nucleus) was under 10%. Correction factor. The counts were corrected using Floedrus' equation Ncor = Nraw x [t/(t + S - 2b)] |
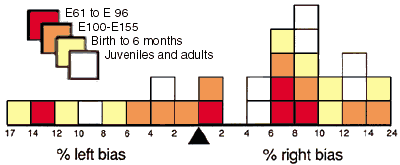
|
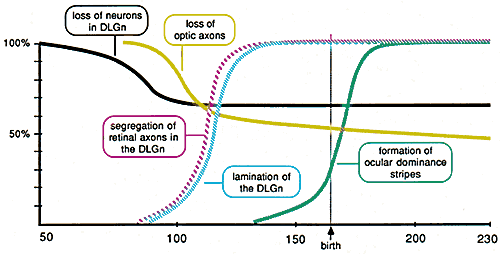
Figure 8. Schematic representation of the timing
of major processes in the development of the primary visual pathway in
the rhesus monkey based on the results of this study, Rakic (1976,
1977a, 1977b), and Rakic and Riley (1983a). A particularly important
observation is that the loss of neurons in the geniculate nucleus (black
line) is essentially complete by the time the number of optic axons in
the nerve begins to fall (gray line). The sensitivity of ganglion cells to a reduction in the number of geniculate neurons is not matched by an equal sensitivity of geniculate neurons to a reduction in the number of ganglion cells. Although the removal of one eye early in development causes a 50% drop in the number of retinal axons innervating the geniculate nuclei, there is no evidence for any substantial reduction in the number of geniculate neurons (P. Rakic and R.W. Williams, in progress). The independence of geniculate neurons to a substantial loss of retinal axons may be due to the major projections the nucleus gets from cortex and brainstem. Neuron death and lamination.The development of cell layers in the geniculate begins around E85 and is complete by about E110 (Fig. 8). If lamination involved the selective loss of neurons, one would anticipate that neuron loss would continue after E100, and one would anticipate that neuron loss would be as common in parvocellular as in magnocellular parts of the nucleus. Neither of these predictions is correct, and consequently, the formation of laminae is evidently not associated with appreciable neuron death. Lamination is probably caused by the growth and redistribution of cells, dendrites, and fibers. Neuron death and the formation of retinotopic mapsThere is good evidence that the formation of retinal projections involves the selective elimination of ganglion cells that make incorrect connections (McLoon, 1982; Insausti et al., 84; Jeffery, 1984; O'Leary et al., 1986; Jacobs et al., 1984, Rakic, 1981; Rakic and Riley, 1983; Williams et al., 1983; Rakic, 1986). In marked contrast, our results demonstrate that neuron number in the geniculate nucleus is stable during the entire period from E110 to E140 when geniculate axons grow into the cortical layers and establish the geniculocortical projection (Fig. 8). It is evident that this precise retinotopic projection is established without the the benefit of cell loss. Thus at the level of retina and its targets, the refinement of connections appears to be achieved in part by the selective elimination of incorrectly connected subsets of ganglion cells, whereas at the level of the geniculate nucleus and its target, the refinement is achieved without any neuron loss. [Footnote 3.] Neuron loss and the development of ocular dominance columnsThe segregation of ocular dominance stripes in the primary visual cortex of the rhesus monkey begins during the last month of gestation and is complete two months after birth (Rakic '76; Hubel et al., 1977; LeVay et al., 1980). LeVay and Stryker (1978) have illustrated arbors of individual Golgi-impregnated geniculocortical axons in neonatal cat that extend uniformly over territories that in adults would occupy several ocular dominance stripes. On the basis of this finding they concluded that segregation results from "synchronous changes in the arborizations of thousands of overlapping geniculocortical axons." Their result does not rule out an alternative, namely, that the refinement is attributable to the selective elimination of geniculate neurons that make incorrect connections. Because, segregation occurs long after the period of neuron death in the geniculate nucleus, we can now rule out this alternative. Positional specificity of neuron lossDegenerating cells in the fetal monkey geniculate nucleus are located principally in ventral and ventro-medial parts of the nucleus. This zone overlaps extensively with the prospective magnocellular layers. In contrast, cell loss is light in the dorsal part of nucleus that gives rise to the four parvocellular layers. There are several possible interpretation of this finding:
Experimental and comparative approaches would be effective in testing the validity of these ideas. Neuron death and the cortical targetThe dependence of young neurons on their target cells has been demonstrated in several systems (reviewed by Hamburger and Oppenheim, 1982; Williams and Herrup, 1988). For instance, in the retinogeniculate system, the reduction in neuron number in the central target—the geniculate nucleus—may initiate a reduction in the number of neurons in retina. This notion that the survival of retinal ganglion cells depends on the number of geniculate neurons stands in contrast to the somatosensory system, in which there is reason to believe that the survival of the central neurons depends on the status of the periphery (Johnson et al., 1972; Rowe, 1982; van der Loos and Welker, 1985). Is the loss of geniculate neurons also regulated by interactions with target cells? The principal targets of these thalamic neurons are cells in striate cortex—stellate cells of layer IV, ascending dendrites of layer V and VI pyramidal cells, and descending dendrites of layer III pyramidal cells (Garey and Powell, 1971; Peters, et al., 1979; White, 1979). At the time the loss begins (ca. E48), only those cortical neurons destined for the deep layers V and VI are being generated, whereas the stellate cells of layer IVC and the pyramidal cells of layer III are not generated until after E66 (Rakic, 1974). All these young neurons migrate up into the cortical plate after passing through a plexus of geniculate axons situated in the optic radiations and the subplate (Rakic, 1977b). While contacts between geniculate axons and migrating neurons during this period have not been demonstrated yet (Kostovic and Rakic, 1980; Chun et al., 1987), it is nonetheless possible that interactions between the two modulate the severity of neuron loss in the thalamus; and it may be more than just coincidental that the period of heaviest neuron death occurs when layer IV neurons migrate through the plexus of geniculate axons. Interactions between thalamic axons and young cortical neurons certainly exercise strong control over the size and number of neurons in the visual cortex of primates. We have been able to demonstrate that reducing the number of geniculate neurons causes a matched reduction in the number of neurons in visual cortex (Rakic and Williams, 1986). Work in progress suggests that the critical period for this type of manipulation extends from E60 to E100, almost precisely the period during which cortical neurons are migrating through the subplate and the plexus of geniculate axons.
ACKNOWLEDGMENTSWe thank Kathryn Ryder for helping us perform the analysis; Dr. Patricia Goldman-Rakic for providing us with tissue; and Dr. Prabhat Sehgal for surgery and acquisition of fetuses of known gestational age. Supported by grant EY 02593 to PR. The fetuses were obtained through the primate breeding colony (Yale University School of Medicine) supported in part by program project NS 22807, and from the New England Regional Primate Research Center, Southboro, MA. LITERATURE CITEDBalado, M., and E. Franke (1937) Das Corpus Geniculatum Externum. Eine anatomisch-klinische Studie. Monographien aus dem Gesamtgebiete der Neurologie und Psychiatrie 62:1–118. Bourgeois, J.-P., and P. Rakic (1983) Synaptogenesis in the primary visual cortex: Quantitative analysis in pre- and postnatal rhesus monkeys. Soc. Neurosci. Abst. 9:692. Caddy, K.W.T., and T.J. Biscoe (1979) Structural and quantitative studies on the normal C3H and Lurcher mutant mouse. Phil Trans. R. Soc. Lond. B. 287:167–201. Chacko, L.W. (1948) The laminar pattern of the lateral geniculate body in the primates. J. Neurol. Neurosurg. Psychiat. 11:211–224. Chalupa, L.M., and R.W. Williams (1985) Formation of retinal projections in the cat. In. R.N. Aslin (ed): Advances in Neuronal and Behavioral Development. vol. 1. Norwood, New Jersey: Ablex Publishing, pp. 1–32. Chow, K.L., J. S. Blum, and R.A. Blum (1950) Cell rations in the thalamo-cortical visual system of Macaca mulatta. J. Comp. Neurol. 92:227–239. Chun, J.J.M., M.J. Nakamura, and C.J. Shatz (1987) Transient cells of the developing mammalian telencephalon are peptide-immunoreactive neurons. Nature 325:617–620. Dineen, J.T., and A.E. Hendrickson (1981) Age-correlated differences in the amount of retinal degeneration after striate cortex lesions in monkeys. Invest. Ophthalmol. Vis. Sci. 21:749–752. Dreher, B., Y. Fukada, and R.W. Rodieck (1976) Identification, classification and anatomical segregation of cells with X-like and Y-like properties in the lateral geniculate nucleus of Old-World primates. J. Physiol. 258:433–452. Garey, L.J., and T.P.S. Powell (1971) An experimental study of the termination of the lateral geniculo-cortical pathway in the cat and monkey. Proc. R. Soc. Lond. (B) 179:41–63. Gundersen, H.J.G. (1977) Notes on the estimation of numerical density of arbitrary profiles. The edge effect. J. Microsc. 111:219–223. Hamburger, V., and R.W. Oppenheim (1982) Naturally occurring neuronal death in vertebrates. Neurosci. Comm. 1: 39–55. Hamori, J., P. Pasik, and T. Pasik (1983) Differential frequency of P-cells and I-cells in magnocellular and parvocellular laminae of monkey lateral geniculate nucleus. An ultrastructural study. Exp. Brain Res. 52:57–66. Hendrickson, A.E., and P. Rakic (1977) Histogenesis and synaptogenesis in the dorsal lateral geniculate nucleus (LGd) of the fetal monkey brain. Anat. Rec. 187:602. Heumann, D., and T. Rabinowicz (1980) Postnatal development of the dorsal lateral geniculate nucleus in the normal and enucleated albino mouse. Exp. Brain Res. 38:75–85. Hubel, D.H., T.N. Wiesel, and S. LeVay (1977) Plasticity of ocular dominance columns in monkey striate cortex. Phil. Trans. R. Soc. Lond. B 278:377–409. Inoue, S. (1986) Video Microscopy. Plenum Press. New York. Insausti, R., C. Blakemore, and W.M. Cowan (1984) Ganglion cell death during development of ipsilateral retino-collicular projections in golden hamster. Nature 308:362–365. Jacobs, D.S., V.H. Perry, and M.J. Hawken (1984) The postnatal reduction of the uncrossed projection from the nasal retina in the cat. J. Neurosci. 4:2425–2433. Jeffery, G. (1984) Retinal ganglion cell death and terminal field retraction in the developing rodent visual system. Dev. Brain Res. 13:81–96. Johnson Jr., J.I., T.C. Hamilton, J-C. Hsung, and P.S. Ulinski (1972) Gracile nucleus absent in adult opossums after leg removal in infancy. Brain Res. 38:421–424. Kaas, J.H., M.F. Huerta, J.T. Weber, and J.K. Harting (1978) Patterns of retinal termination and laminar organization of the lateral geniculate nucleus of primates. J. Comp. Neurol. 182:517–554. Konigsmark, B.W. (1970) Methods for the counting of neurons. In: W.J.H. Nauta and S.O.E. Ebbesson, Contemporary Research Methods in Neuroanatomy. New York, Springer-Verlag. pp.315–340. Kostovic, I.M, and P. Rakic (1980) Cytology and time of origin of interstitial neurons in the white matter in infant and adult human and monkey telencephalon. J. Neurocytol. 9:219–242. Kostovic, I.M., and P. Rakic (1984) Development of prestriate visual projections in the monkey and human fetal cerebrum revealed by transient cholinesterase staining. J. Neurosci. 4:25–42. Kupfer, C., L. Chumbley, and J. De C. Downer (1967) Quantitative histology of optic nerve, optic tract and lateral geniculate nucleus of man. J. Anat. 101:393–401. LaVail, M.M., D. Yasumura, and P. Rakic (1983) Cell genesis in the rhesus monkey retina. Invest. Ophthalmol. Vis. Sci. 24:7. LeVay, S., and M.P. Stryker (1978) The development of ocular dominance columns in the cat. In: Aspects of Developmental Neurobiology, ed. J. A. Ferrendelli. Soc. Neurosci. Symp. 4:83–98. LeVay, S., T.N. Wiesel, and D.H. Hubel (1980) The development of ocular dominance columns in normal and visually deprived monkeys. J. Comp. Neurol. 191:1–51. Levitt, P., and P. Rakic (1980) Immunoperoxidase localization of glial fibrillary acid protein in radial glial cells and astrocytes of the developing rhesus monkey brain. J. Comp. Neurol. 193: 815–840. Levitt, P., Cooper, M.L., and Rakic, P. (1983) Early divergence and changing proportion of neuronal and glial precursor cells in the primate cerebral ventricular zone. Dev. Biol. 96:427–484. Ling, E.A, J.A. Paterson, A. Privat, S. Mori , and C.P. Leblond (1973) Investigation of glial cells in semithin sections. I. Identification of glial cells in the brains of young rats. J. Comp. Neurol. 149:43–72. Malpeli, J.G., and F.H. Baker (1975) The representation of the visual field in the lateral geniculate nucleus of Macaca mulatta. J. Comp. Neurol. 161:569–594. Marrocco, R.T., J.W. McClurkin, and R.A. Young (1982) Spatial summation and conduction latency classification of cells of the lateral geniculate nucleus of macaques. J. Neurosci. 2:1275–1291. Matthews, M.A., C.H. Narayanan, Y. Narayanan, and D.J. Siegenthaler-Matthews (1982) Inhibition of axoplasmic transport in the developing visual system of the rat. III. Electron microscopy and Golgi studies of retino-fugal synapses and post-synaptic neurons in the dorsal lateral geniculate nucleus. Neuroscience 7:405–422. McLoon, S.C. (1982) Alteration in precision of the crossed retinotectal projection during chick development. Science 215:1418–1420. Montero, V.M., and J. Zempel (1986) The proportion and size of GABA-immunoreactive neurons in the magnocellular and parvocellular layers of the lateral geniculate nucleus of the rhesus monkey. Exp. Brain Res. 62:215–223. Nishimura, Y., and P. Rakic (1985) Development of the rhesus monkey retina. I. Emergence of the inner plexiform layer and its synapses. J. Comp. Neurol. 241:420–434. Nishimura, Y., and P. Rakic (1987) Development of the rhesus monkey retina: II. A three-dimensional analysis of the sequence of synaptic combinations in the inner plexiform layer. J. Comp. Neurol. XX:XXX–XXX. Norden, J.J., and J.H. Kaas (1978) The identification of relay neurons in the dorsal lateral geniculate nucleus of monkeys using horseradish peroxidase. J. Comp. Neurol. 182:707–725. Ogren, M.P., P. Rakic, and P. Goldman-Rakic (1984) Consequences of prenatal striate cortex lesions on retinogeniculate projections in the monkey. Invest. Ophthalmol. Vis. Sci. 24:64. O'Leary, D.D.M., J.W. Fawcett, and W.M. Cowan (1986) Topographic targeting errors in the retinocollicular projection and their elimination by selective ganglion cell death. J. Neurosci. 6:3692–3705. Pasik, P., T. Pasik, J. Hamori, and G.R. Holstein (1986) GABA immunoreactivity in monkey lateral geniculate nucleus (LGN) at light and electron microscopic levels. Soc. Neurosci. Abs. 11:317. Perry, V.H., R. Oehler, and A. Cowey (1984) Retinal ganglion cells that project to the dorsal lateral geniculate nucleus in the macaque monkey. Neuroscience 12:1101–1123. Peters, A., C.C. Proskauer, M.L. Feldman, and L. Kimerer (1979) The projection of lateral geniculate nucleus to area 17 of the rat cerebral cortex. V. Degenerating axon terminals synapsing with Golgi-impregnated neurons. J. Neurocytol. 8:331–357. Prestige, M.C. (1970) Differentiation, degeneration, and the role of the periphery: Quantitative considerations. In The Neurosciences, Second Study Program F.O. Schmitt, ed. pp. 73–82. New York. Rockerfeller Univ. Press. Provis, J.M., D. van Driel, F.A. Billson, and P. Russell (1985) Human fetal optic nerve: Overproduction and elimination of retinal axons during development. J. Comp. Neurol. 238:92–101. Rakic, P. (1972) Mode of cell migration to the superficial layers of fetal monkey neocortex. J. Comp. Neurol. 145:61–84. Rakic, P. (1974) Neurons in rhesus monkey visual cortex: Systematic relation between time of origin and eventual disposition. Science: 183:425–427. Rakic, P. (1976) Prenatal genesis of connections subserving ocular dominance in the rhesus monkey. Nature 261:467–471. Rakic, P. (1977a) Genesis of the dorsal lateral geniculate nucleus in the rhesus monkey: Site and time of origin, kinetics of proliferation, routes of migration and pattern of distribution of neurons. J. Comp. Neurol. 176:23–52. Rakic, P. (1977b) Prenatal development of the visual system in rhesus monkey. Phil Trans. R. Soc. Lond. B. 278:245–260. Rakic, P. (1981) Development of visual centers in the primate brain depends on binocular competition before birth. Science 214:928–931. Rakic, P. (1986) Mechanisms of ocular dominance segregation in the lateral geniculate nucleus: Competitive elimination hypothesis. Trends Neurosci. 5:11–19. Rakic, P., and K.P. Riley (1983a) Overproduction and elimination of retinal axons in the fetal rhesus monkey. Science 219:1441–1444. Rakic, P., and K.P. Riley (1983b) Regulation of axon number in primate optic nerve by prenatal binocular competition. Nature 305:135–137. Rakic, P., and R.W. Williams (1986) Thalamic regulation of cortical parcellation: An experimental perturbation of the striate cortex in rhesus monkeys. Soc. Neurosci. Abst. 12:1499. Rowe, M.J. (1982) Development of mammalian somatosensory pathways. Trends Neurosci. 5:408–411. Satorre, J., J. Cano, F. Reinoso-Suárez (1986) Quantitative cellular changes during postnatal development of the rat dorsal lateral geniculate nucleus. Anat. Embryol. 174:321–327. Schiller P.H., and J.G. Malpeli (1978) Functional specificity of lateral geniculate nucleus laminae of the rhesus monkey. J. Neurophysiol. 41:788–797. Scholtz, C.L., K. Swettenham, A. Brown, and D.M.A. Mann (1981) A histoquantitative study of the striate cortex and lateral geniculate body in normal, blind and demented subjects. Neuropath. Appl. Neurobiol. 7:103–114. Sengelaub, D.R., L.F. Jacobs, and B.L. Finlay (1985) Regional differences in normally occurring cell death in the developing hamster lateral geniculate nuclei. Neurosci. Let. 55:103–108. Shapley, R., E. Kaplan, and R. Soodak (1981) Spatial summation and contrast sensitivity of X and Y cells in the lateral geniculate nucleus of the macaque. Nature 292:543–545. Shatz, C.J., and M.B. Luskin (1986) The relationship between the geniculocortical afferents and their cortical target cells during development of the cat's primary visual cortex. J. Neurosci. 6:3655–3668. Shatz, C.J., and P. Rakic (1981) The genesis of efferent connections from the visual cortex of the fetal rhesus monkey. J. Comp. Neurol. 196:287–307. Shatz, C.J., and D. W. Sretavan (1986) Interactions between retinal ganglion cells during the development of the mammalian visual system. Ann. Rev. Neurosci. 9: 171–207. Shotwell, S.L., C.J. Shatz, and M.B. Luskin (1986) Development of glutamic acid decarboxylase immunoreactivity in the cat's lateral geniculate nucleus. J. Neurosci. 6:1410–1423. Sullivan, P.R., J. Kuten, M.S. Atkinson, J.B. Angevine, and P.I. Yakovlev (1958) Cell count in the lateral geniculate nucleus of man. Neurol. 58: 566–567 van der Loos, H., and E. Welker (1985) Development and plasticity of somatosensory brain maps. In: M.J. Rowe and W.D. Willis (eds): Development, Organization, and Processing in Somatosensory Pathways. New York: Alan Liss. Weller, R. E., J.H. Kaas, and A.B. Wetzel (1979) Evidence for the loss of X-cells of the retina after long-term ablation of visual cortex in monkeys. Brain Res. 160:134–138. White, E.L. (1979) Thalamocortical synaptic relations: A review with emphasis on the projections of specific thalamic nuclei to the primary sensory areas of the neocortex. Brain Res. Rev. 1:275–311. Williams, R.W., M.J. Bastiani, and L.M. Chalupa (1983) Loss of axons in the cat optic nerve following fetal unilateral enucleation: An electron microscopic analysis. J. Neurosci. 3:1554–1564. Williams, R.W., M.J. Bastiani, B.Lia, and L.M. Chalupa (1986) Growth cones, dying axons, and developmental fluctuations in the fiber population of the cat's optic nerve. J. Comp. Neurol. 246:32–69. Williams, R.W., and K. Herrup (1988) Control of neuron number. Ann. Rev. Neurosci. 11:423–453. Williams, R.W., and P. Rakic (1985) Number of neurons in the monkey's dorsal lateral geniculate nucleus during development. Soc. Neurosci. Abs. 11:804. Williams, R.W., and P. Rakic (1986) Pronounced architectonic differences between monocular and binocular segments of the monkeys striate cortex. Soc. Neurosci. Abst. 12:1498. Williams, R.W., K. Ryder, and P. Rakic (1987) Emergence of cytoarchitectonic differences between areas 17 and 18 in the developing rhesus monkey. Soc. Neurosci. Abst. 13:in press. Yakovlev, P.I. (1970) Whole brain serial histological sections. In:
Neuropathology: Methods and Diagnosis. C.G. Tedeschi. Little, Brown and
Company, 1970, pp.371–378.
Footnote 2. Both humans and rhesus macaques have 1.2 to 1.5 million retinal ganglion cells and about 1.0 to 1.5 million geniculate neurons (Chacko, 1948; Chow et al., 1950; Sullivan et al., 1958). Developmental changes in the number of retinal ganglion cell axons are also remarkably similar in these two primates (cf., Rakic and Riley, 1983; Provis et al., 1986). Using the gestational age at which retinal axon number peaks in the two species as a time standard (E90 in rhesus and E110 in human), we estimate that the putative period of neuron loss in the human lateral geniculate should extend from E60 to E120. Gestation is 165 days in rhesus monkeys and 265 days in humans. Footnote 3. A template of geniculocortical topography could conceivably be established between geniculate axons and cells in the subplate before the geniculate axons grow into the cortex. If this is the case then the subplate may play a role in organizing or filtering axons destined for visual cortex. |
Since 11 August 98, Updated 21 January 2001